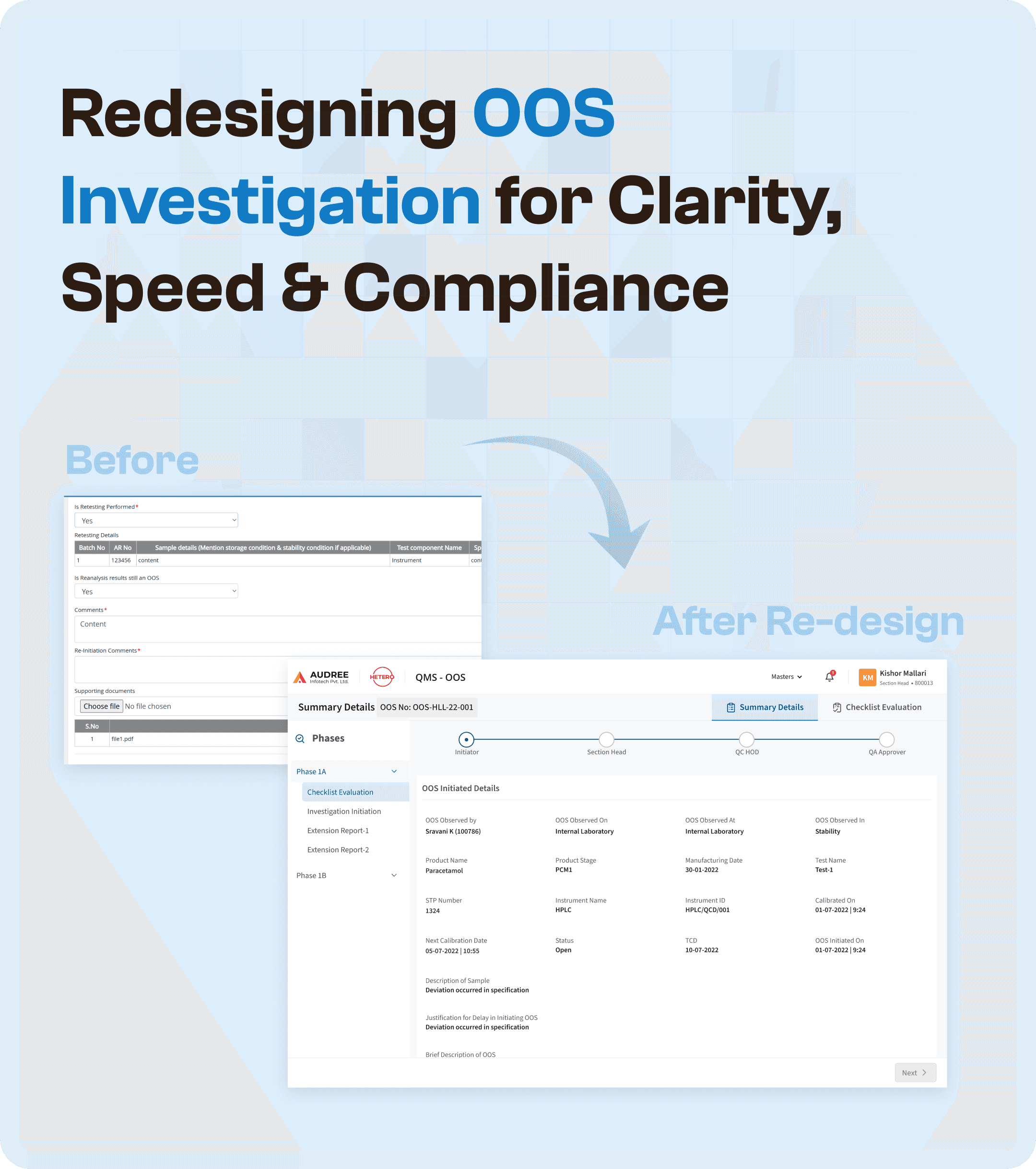

The Core Problem: A Fragmented OOS Process
The initial OOS investigation process had several pain points that made it difficult for teams to effectively manage investigations:
Cluttered Forms & Repeated Entries: Investigators had to input the same information multiple times, causing delays and increasing the chances of errors.
Slow and Delayed Closures: The lack of clear ownership and process stages made it difficult for teams to close investigations in a timely manner.
Difficulty Validating Entries: With information spread across different systems and forms, it was hard to track previous steps and verify whether the investigation was progressing as expected.
Additionally, teams heavily relied on manual coordination between departments like QA, QC, and production teams. This not only added more time to the process but also created confusion due to inconsistent data and communication.

Our Goal: A Guided, Predictable Workflow
The goal of this redesign was to turn a complex, multi-step investigation process into a structured, role-based workflow that is:
Faster: Speed up investigation closures by creating a predictable process that teams can follow at each stage.
More Traceable: Provide clear visibility at each phase of the investigation, making it easier for teams to track progress and ensure regulatory compliance.
Less Manual Effort: Reduce manual data entry, streamline communication, and improve coordination between departments.
By focusing on these objectives, we aimed to create a more efficient and compliant OOS investigation workflow that would lead to faster and more accurate decision-making.

Our UX Approach: Mapping Every Investigation Phase
The OOS investigation process involves several key stages, including identifying out-of-specification results, investigating the causes, and determining corrective actions. We began by mapping all investigation phases end-to-end to identify inefficiencies, redundancies, and areas of confusion.
Here’s how we tackled the redesign:
1. Mapping the Investigation Phases
Each stage of the investigation was mapped out in detail to provide clear structure and ownership. The goal was to ensure that every department involved had clear visibility of the tasks at hand, as well as who was responsible for each action.
2. Rebuilding the Lifecycle
The OOS investigation lifecycle was restructured into clear, role-based steps, with clearly defined inputs, actions, and responsibilities at each stage. This eliminated confusion and helped streamline the overall process.
3. Reducing Confusion and Backtracking
By removing redundant steps and improving navigation, the new workflow helps reduce back-and-forth between departments. Each phase is now clearly defined with no overlapping tasks, ensuring smooth transitions between stages.

Design Highlights: A Structured, Easy-to-Follow Interface
We redesigned the OOS investigation interface with user clarity and simplicity in mind. One of the key highlights of the new design is the phase-wise view, which offers clear status tags, structured evidence, and visibility into previous steps. Here’s how we made it more intuitive:
Structured Investigation Phases: Each phase of the OOS investigation is now displayed in a clean, organized layout. The clear status tags help teams easily identify where they are in the process and what actions are needed to move to the next phase.
Smarter Action Cues: The design also incorporates visual cues to indicate the next step in the process, ensuring that teams know exactly what to do next without wasting time.
Increased Visibility: Users now have clear insights into the investigation’s progress, with easy-to-read summaries and detailed evidence that can be accessed at any time. This ensures a transparent workflow for all departments.

Why OOS Investigations Matter in Pharma
Out-of-specification (OOS) investigations are an essential part of pharmaceutical quality control. When a product does not meet the required specifications, it’s vital to quickly identify the cause, take corrective actions, and prevent future occurrences. A streamlined, well-documented investigation process is crucial for:
Ensuring Product Safety: Accurate and timely OOS investigations ensure that only safe, effective products reach consumers.
Maintaining Regulatory Compliance: Pharmaceutical companies must adhere to strict regulatory requirements, and a structured OOS process helps ensure compliance with industry standards.
Improving Operational Efficiency: By reducing inefficiencies in the investigation process, companies can save time, reduce costs, and ensure better resource utilization.
Impact: Faster Investigations with Less Manual Effort
The redesigned OOS process has already led to significant improvements in the speed and accuracy of investigations:
Faster Task Completion: The structured, guided workflow has drastically reduced the time needed to complete each investigation, resulting in quicker decision-making and faster closure of cases.
Less Manual Effort: By automating certain steps and reducing repetitive data entry, the new system requires less manual effort, freeing up team members to focus on more critical tasks.
Improved Compliance: The clear visibility and structured approach have improved traceability across the investigation process, ensuring that every action is documented and aligned with regulatory requirements.



