
Introduction: The Challenge of Quality Agreement Management
In pharmaceutical industries, Quality Agreements define responsibilities and quality expectations between pharma companies and their partners. However, managing these agreements is no small feat. The process often involves multiple stakeholders, such as quality control (QC) teams, quality assurance (QA), and supply chain personnel.
The traditional approach to managing Quality Agreements typically relies on outdated, fragmented software systems. These systems are cluttered, hard to navigate, and lack clear visibility into each agreement's stage. Teams frequently rely on manual reviews, which causes delays and hampers progress.
Pharma teams spent excessive time coordinating between emails, documents, and scattered screens. This back-and-forth process led to confusion, misunderstandings, and longer approval cycles. As a result, manual coordination and lack of clarity became significant roadblocks in the workflow.

The Problem: Fragmented Workflows and Inefficiencies
The root cause of these inefficiencies lay in how the Quality Agreement lifecycle was structured in the existing system. Some key problems included:
Manual Reviews: Teams had to manually review clauses, cross-reference documents, and check for inconsistencies across multiple systems.
Scattered Screens: Information was spread across multiple screens, leading to confusion and unnecessary navigation.
Scattered Communication: Teams relied heavily on offline communication (emails, phone calls) to verify changes and confirm approvals.
These issues resulted in slow and inefficient workflows, causing significant delays in the approval process for Quality Agreements.

The Goal: Simplifying and Streamlining the Quality Agreement Lifecycle
The goal of the redesign was clear: to simplify and modernize the Quality Agreement process, eliminating redundancies and ensuring better workflow clarity and transparency.
Our objectives were:
Clearer Clause Grouping: Improve how clauses are grouped, allowing for easier navigation and visibility.
Stronger Traceability: Provide transparency into every stage of the Quality Agreement lifecycle.
Reduced Manual Coordination: Minimize back-and-forth communication and manual checks by automating stages and providing clear, actionable steps.
Our approach focused on streamlining the review process and making it easier for teams to track progress and complete tasks with minimal effort.

The UX Approach: Turning a Scattered Process into a Streamlined System
After thoroughly analyzing the existing workflow, we identified opportunities for improvement and began transforming the Quality Agreement process into a more efficient, user-friendly experience.
Key Changes in the UX Approach:
Unified Workflow: The first major step was to unify the fragmented process into a single, structured workflow. This eliminated the need for switching between screens and documents, providing visibility at every stage.
Full Visibility: We introduced clear progress tracking and status indicators to help teams understand where each agreement was in the process and what steps remained. No more jumping between PDFs, emails, and screens. Everything was now integrated into a single interface.
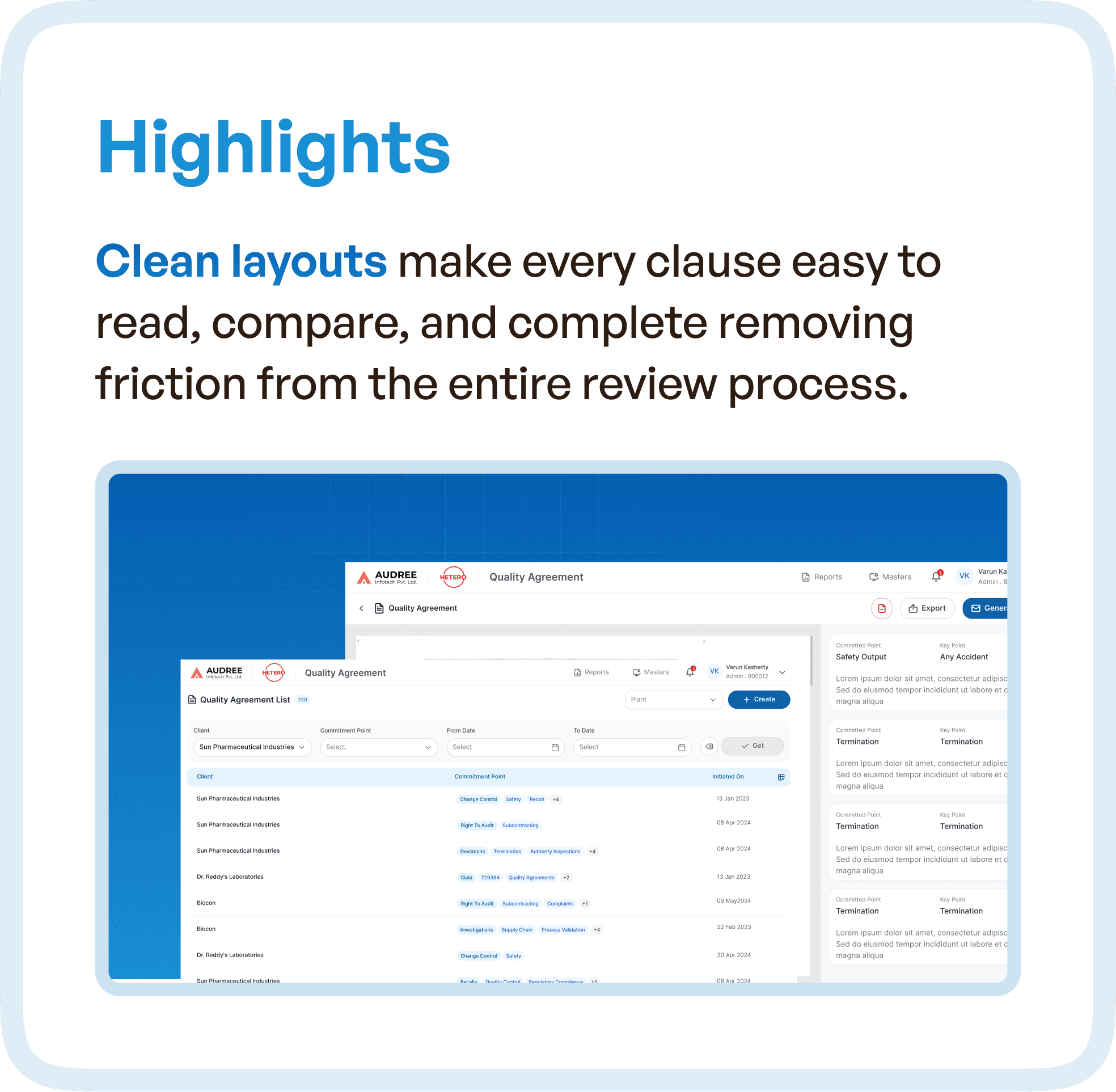
Optimized Review Process: We created clean layouts that made reviewing individual clauses easy and intuitive. By grouping clauses logically and offering tools to compare revisions, teams could now review agreements with greater clarity and confidence.

Design Highlights: Clean Layouts and Intuitive Navigation
A significant part of the redesign was focused on improving the visual hierarchy and making the system more intuitive to navigate.
We used clean layouts that helped users quickly access relevant information without feeling overwhelmed. Key features included:
Clause Grouping: Clauses were grouped logically to reduce visual clutter and ensure that users could find specific information faster.
Progress Tracking: A clear visual representation of each stage in the process, enabling users to see at a glance what was completed and what remained.
Version Tracking: Version history and comparisons allowed teams to track changes and easily identify what was modified in previous drafts.
The redesign also introduced structured views for each clause, making it easier for users to compare and make edits. This minimized confusion and reduced the time spent on manual checks.
Improved Communication: No More Back-and-Forth
One of the most important goals of the redesign was to improve communication among teams and reduce manual coordination.
In the previous system, communication gaps often led to delays in the approval process. Teams were forced to exchange emails, make phone calls, and rely on offline methods to verify changes.
In the redesigned system, we introduced built-in comments and approval tracking. Users could now:
Leave feedback directly on the system for specific clauses or sections.
Track comments and approvals in real-time, eliminating the need for back-and-forth emails.
Clearly see what has been approved and what requires attention, making the entire process more transparent.
These changes enabled teams to communicate efficiently within the platform, speeding up the review process and reducing unnecessary delays.
The Impact: Faster Reviews and Increased Confidence in the Process
The redesigned system led to several notable improvements:
Faster Approvals: With the new interface, teams could review and approve Quality Agreements faster, thanks to streamlined navigation and automated tracking.
Less Manual Effort: With fewer manual steps required, teams were able to focus on the more critical aspects of the review process rather than worrying about missing data or untracked changes.
Increased Confidence: The system’s transparency and clarity allowed teams to feel more confident in the process, knowing that the system was organized and that all relevant changes were tracked.
Ultimately, the new design enabled pharma teams to work more efficiently, reduce bottlenecks, and deliver better Quality Agreements with fewer errors.

Why Quality Agreement UX Redesign Matters for Pharma
For pharmaceutical companies, quality agreements are essential to ensuring compliance and maintaining effective partnerships with other stakeholders. An inefficient process, especially one that relies on outdated software or manual coordination, can cause significant delays and compliance risks.
Redesigning these systems with a focus on user experience can streamline the approval process, improve collaboration across teams, and ultimately enhance the speed and accuracy of Quality Agreement reviews.
This case study highlights the importance of UX optimization in complex systems, particularly in the pharmaceutical industry. By focusing on clarity, transparency, and better communication, UX-driven redesigns can transform outdated, inefficient processes into streamlined workflows that boost productivity and reduce errors.
How Upslide Design Studio Optimizes Pharma Software UX
At Upslide Design Studio, we specialize in enterprise UX design, focusing on systems like Quality Agreement management for pharma. Whether it’s LIMS, BMR, or NDIR systems, we help organizations streamline their workflows and improve the user experience.
Our process includes:
User research to understand the needs of all stakeholders
Mapping workflows to identify pain points and inefficiencies
UX redesigns that focus on simplifying and clarifying complex processes
Testing to ensure the new design is both effective and intuitive
By focusing on real user needs and ensuring that every step in the workflow is clear and connected, we help organizations optimize their systems for better results.
Final Thought: Redesigning Quality Agreements for Efficiency
The case study of the Quality Agreement UX redesign shows that UX optimization is not just about improving aesthetics. It’s about creating a clear, efficient, and collaborative process that empowers teams to work faster and more confidently.
If your Quality Agreement system is outdated, scattered, or relies too heavily on manual communication, it’s time for a redesign. At Upslide Design Studio, we can help transform your system into a guided, user-friendly platform that ensures faster approvals and fewer errors, resulting in a more efficient review process.


