Streamlining New Drug Documentation
for Smarter Workflows
Streamlining New Drug Documentation
for Smarter Workflows
Streamlining New Drug Documentation
for Smarter Workflows
About Project
About Project
About Project
NDIR (New Drug Introduction and Registration) is a pharmaceutical software that manages the complete lifecycle of new drug documents from initiation and creation to approval, revision, and obsolescence.
Audree Infotech, one of India’s leading pharmaceutical software companies, identified the need to enhance NDIR’s usability and efficiency. The previous system required heavy manual input, lacked clarity in tracking document stages, and made version management complex for users.
Our objective was to simplify and modernize the NDIR experience enabling pharma teams to manage document lifecycles seamlessly, reduce manual effort, and ensure complete visibility and control across every stage.
NDIR (New Drug Introduction and Registration) is a pharmaceutical software that manages the complete lifecycle of new drug documents from initiation and creation to approval, revision, and obsolescence.
Audree Infotech, one of India’s leading pharmaceutical software companies, identified the need to enhance NDIR’s usability and efficiency. The previous system required heavy manual input, lacked clarity in tracking document stages, and made version management complex for users.
Our objective was to simplify and modernize the NDIR experience enabling pharma teams to manage document lifecycles seamlessly, reduce manual effort, and ensure complete visibility and control across every stage.
About Project
Pharmaceutical
Team
Anush Reddy, S.Madhumala
Subscription Category
Quick win
Project start Year
November 2024
Core Business Challenges
Core Business Challenges
Core Business Challenges
Poor User Adoption Due to Outdated Experience
Poor User Adoption Due to Outdated Experience
Poor User Adoption Due to Outdated Experience
The outdated interface and scattered workflows made the system hard to use, lowering adoption across departments.
The outdated interface and scattered workflows made the system hard to use, lowering adoption across departments.
The outdated interface and scattered workflows made the system hard to use, lowering adoption across departments.
Stuck in a Loop, Slowed by Inefficiency
Stuck in a Loop, Slowed by Inefficiency
Stuck in a Loop, Slowed by Inefficiency
Repeated support tickets for the same issues slowed teams down and overloaded QA, all because document progress wasn’t clear.
Repeated support tickets for the same issues slowed teams down and overloaded QA, all because document progress wasn’t clear.
Repeated support tickets for the same issues slowed teams down and overloaded QA, all because document progress wasn’t clear.
Heavy Dependence on Manual Coordination
Heavy Dependence on Manual Coordination
The manual, multi-step document lifecycle slowed approvals and revisions, delaying overall new-drug documentation readiness.
The manual, multi-step document lifecycle slowed approvals and revisions, delaying overall new-drug documentation readiness.
Heavy Dependence on Manual Coordination
The manual, multi-step document lifecycle slowed approvals and revisions, delaying overall new-drug documentation readiness.
Our Approach
Our Approach
Our Approach
Mapping User Flows to Uncover Hidden Gaps
We mapped end-to-end user journeys from document initiation to revision and obsolescence to identify redundant steps and friction points. Streamlined flows ensured users could move through each stage without backtracking or confusion.
Mapping User Flows to Uncover Hidden Gaps
We mapped end-to-end user journeys from document initiation to revision and obsolescence to identify redundant steps and friction points. Streamlined flows ensured users could move through each stage without backtracking or confusion.
Mapping User Flows to Uncover Hidden Gaps
We mapped end-to-end user journeys from document initiation to revision and obsolescence to identify redundant steps and friction points. Streamlined flows ensured users could move through each stage without backtracking or confusion.
Designing a Seamless, Traceable, and Efficient NDIR Experience
Designing a Seamless, Traceable, and Efficient NDIR Experience
Designing a Seamless, Traceable, and Efficient NDIR Experience
We redesigned the complete NDIR workflow from initiation to approval, revision, and obsolescence into clear, guided stages. Each step was simplified to reduce effort, remove redundancies, and make progress easy to track. With structured approvals, transparent revisions, and clear status visibility, users always know what’s done and what comes next.
We redesigned the complete NDIR workflow from initiation to approval, revision, and obsolescence into clear, guided stages. Each step was simplified to reduce effort, remove redundancies, and make progress easy to track. With structured approvals, transparent revisions, and clear status visibility, users always know what’s done and what comes next.




Simplified Document Initiation Form
Simplified Document Initiation Form
Simplified Document Initiation Form
We simplified the document initiation process by bringing all key fields product details, document type, and customer data into one structured form. With a clear hierarchy and collapsible sections, users can easily upload documents, add comments, and switch between initiation, revision, or addendum making submissions faster and error-free.
We simplified the document initiation process by bringing all key fields product details, document type, and customer data into one structured form. With a clear hierarchy and collapsible sections, users can easily upload documents, add comments, and switch between initiation, revision, or addendum making submissions faster and error-free.
We simplified the document initiation process by bringing all key fields product details, document type, and customer data into one structured form. With a clear hierarchy and collapsible sections, users can easily upload documents, add comments, and switch between initiation, revision, or addendum making submissions faster and error-free.
Simplified Change Reviews for clarity
Simplified Change Reviews for clarity
Simplified Change Reviews for clarity
We refined the Change Request Review screen to give users a clear view of document revisions in one place. Organized sections for details, actions, and closure make it easy to review, upload updates, and add comments—ensuring faster collaboration and approvals.
We refined the Change Request Review screen to give users a clear view of document revisions in one place. Organized sections for details, actions, and closure make it easy to review, upload updates, and add comments—ensuring faster collaboration and approvals.
We refined the Change Request Review screen to give users a clear view of document revisions in one place. Organized sections for details, actions, and closure make it easy to review, upload updates, and add comments—ensuring faster collaboration and approvals.


Seamless Navigation Through Adaptive Components
Seamless Navigation Through Adaptive Components
Seamless Navigation Through Adaptive Components
We designed adaptable components to ensure clarity across all stages. These reusable elements simplify recognition, reduce confusion, and keep the interface consistent and scalable.
We designed adaptable components to ensure clarity across all stages. These reusable elements simplify recognition, reduce confusion, and keep the interface consistent and scalable.








The New NDIR Experience Simplified and Seamless
The New NDIR Experience Simplified and Seamless
The New NDIR Experience Simplified and Seamless
We redesigned the NDIR interface for clarity and ease, enabling pharma teams to manage document initiation, revision, and approvals faster and more efficiently.
We redesigned the NDIR interface for clarity and ease, enabling pharma teams to manage document initiation, revision, and approvals faster and more efficiently.



Result That Transformed NDIR Lifecycle
Result That Transformed NDIR Lifecycle
Result That Transformed NDIR Lifecycle
Connected workflows, structured stages, and a fully digital execution model simplified new drug documentation and improved coordination across global teams.
Connected workflows, structured stages, and a fully digital execution model simplified new drug documentation and improved coordination across global teams.

Stronger Traceability & Control
Stronger Traceability & Control
Stronger Traceability & Control
Structured document stages, standardized components, and clear status indicators ensured every initiation, revision, and approval was fully traceable across departments.
Structured document stages, standardized components, and clear status indicators ensured every initiation, revision, and approval was fully traceable across departments.
Structured document stages, standardized components, and clear status indicators ensured every initiation, revision, and approval was fully traceable across departments.

Faster Document Approvalsption
Guided approval workflows replaced email-based coordination and manual tracking. Review cycles became predictable and efficient, enabling up to 40% faster approvals across critical documents.

Fewer Support Tickets
Clear status visibility, role-based actions, and in-app notifications reduced user confusion. As a result, teams saw significantly fewer support requests and smoother cross-functional collaboration.

Faster Document Processing
Faster Document Processing
Guided workflows and tab-based navigation replaced fragmented manual steps, enabling faster NDIR processing from initiation to approval.
Guided workflows and tab-based navigation replaced fragmented manual steps, enabling faster NDIR processing from initiation to approval.

Fewer Support Tickets
Fewer Support Tickets
Clear ownership, consistent statuses, and in-app visibility reduced follow-ups, enabling smoother cross-team collaboration across global operations.
Clear ownership, consistent statuses, and in-app visibility reduced follow-ups, enabling smoother cross-team collaboration across global operations.
Deep-Dive Into More System
Deep-Dive Into More System
Deep-Dive Into More System
Browse every optimised Software and explore how legacy systems became intuitive.
Browse every optimised Software and explore how legacy systems became intuitive.
RCAI

Root Cause Analysis with Intelligence
LMS
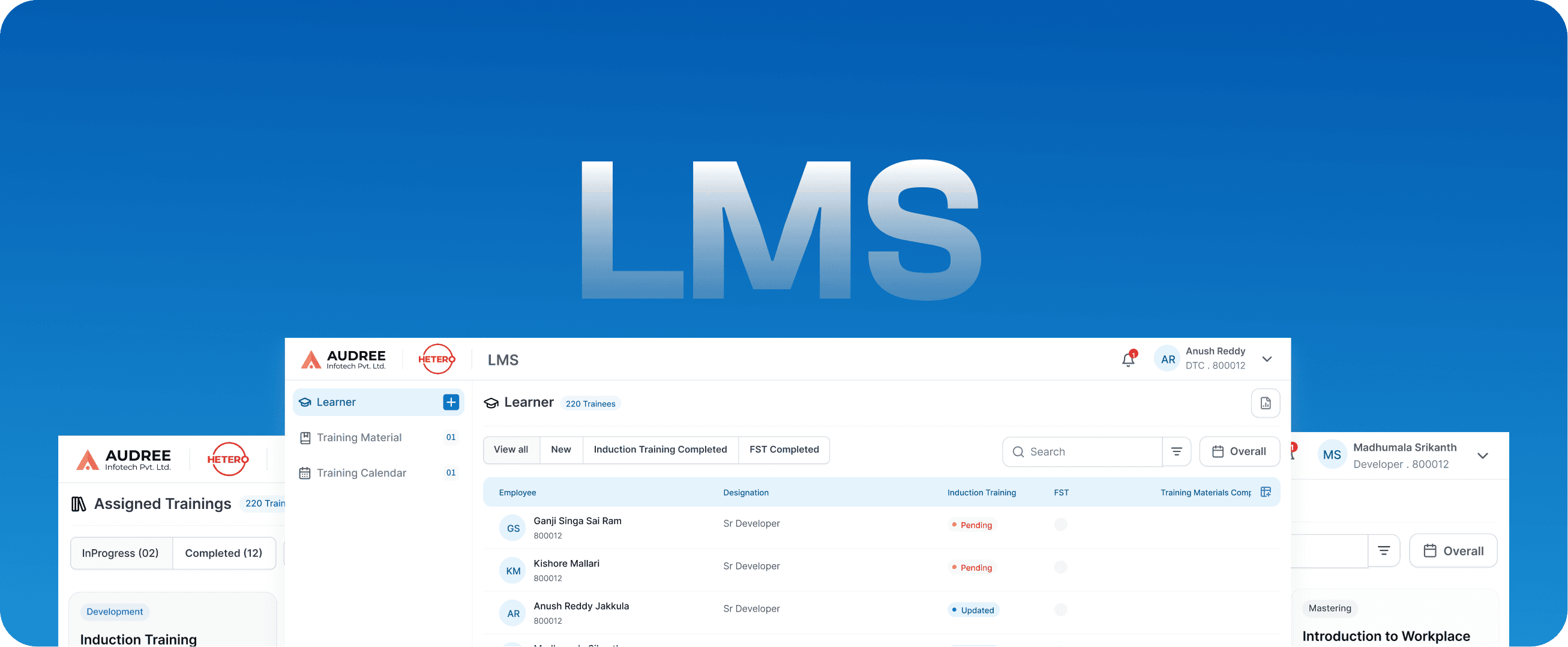
Learning Management System
LIMS
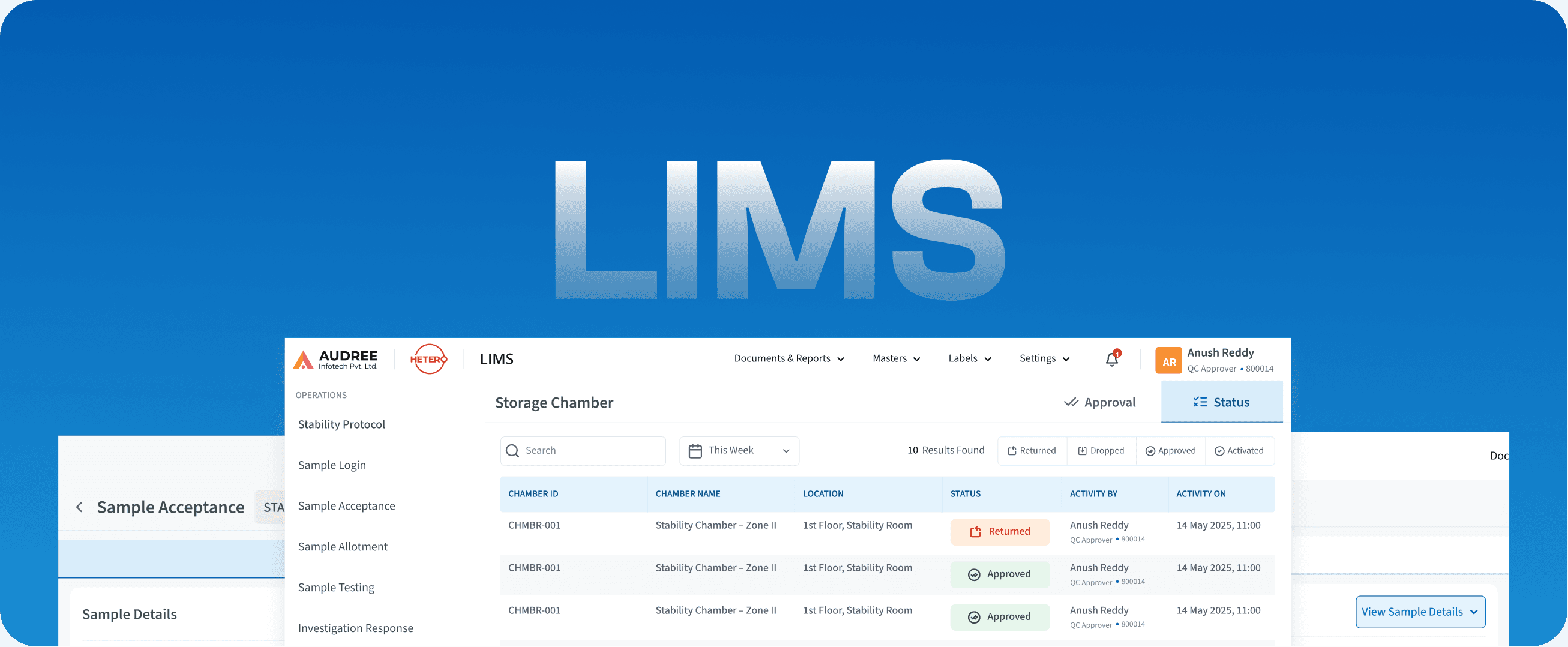
Laboratory Information Management System
S & OP
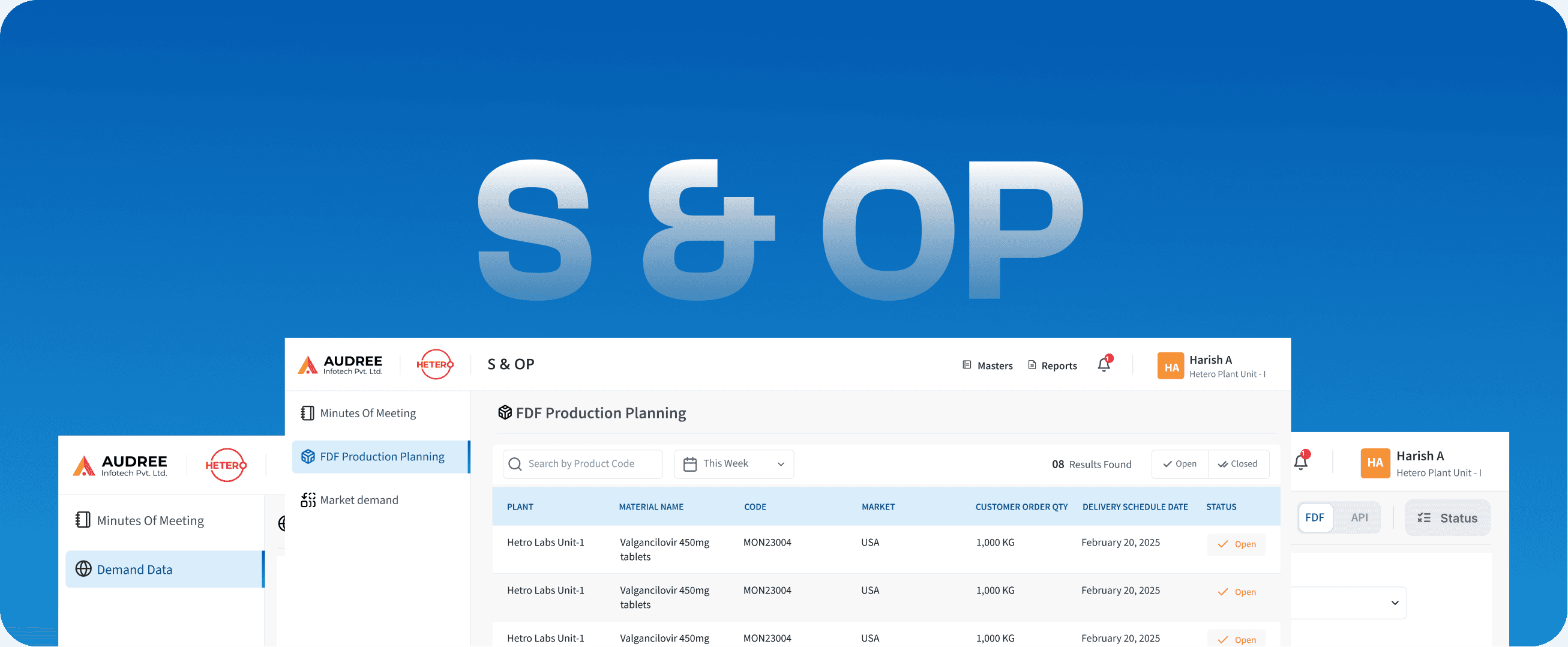
Sales & Operations Planning
E-BMR
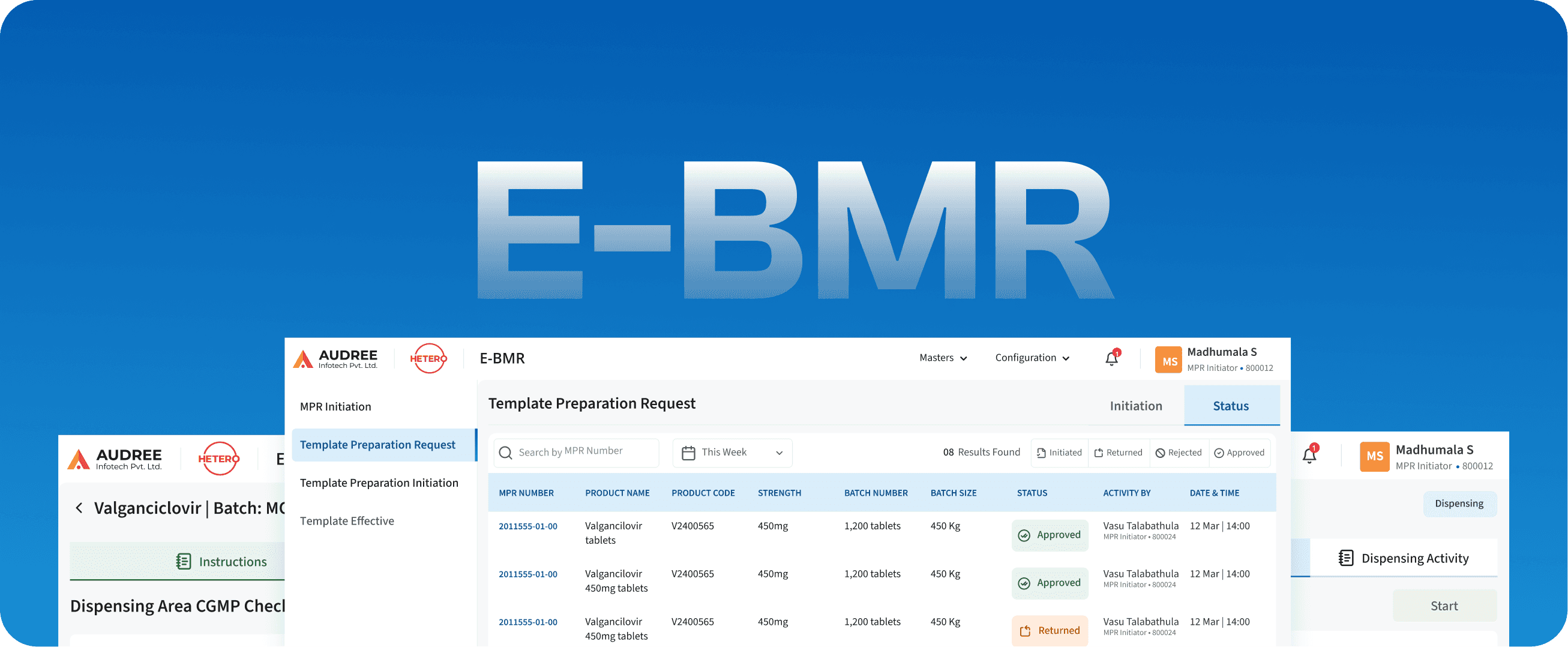
Batch Manufacturing Recall
WMPS

Warehouse Management System
DMS
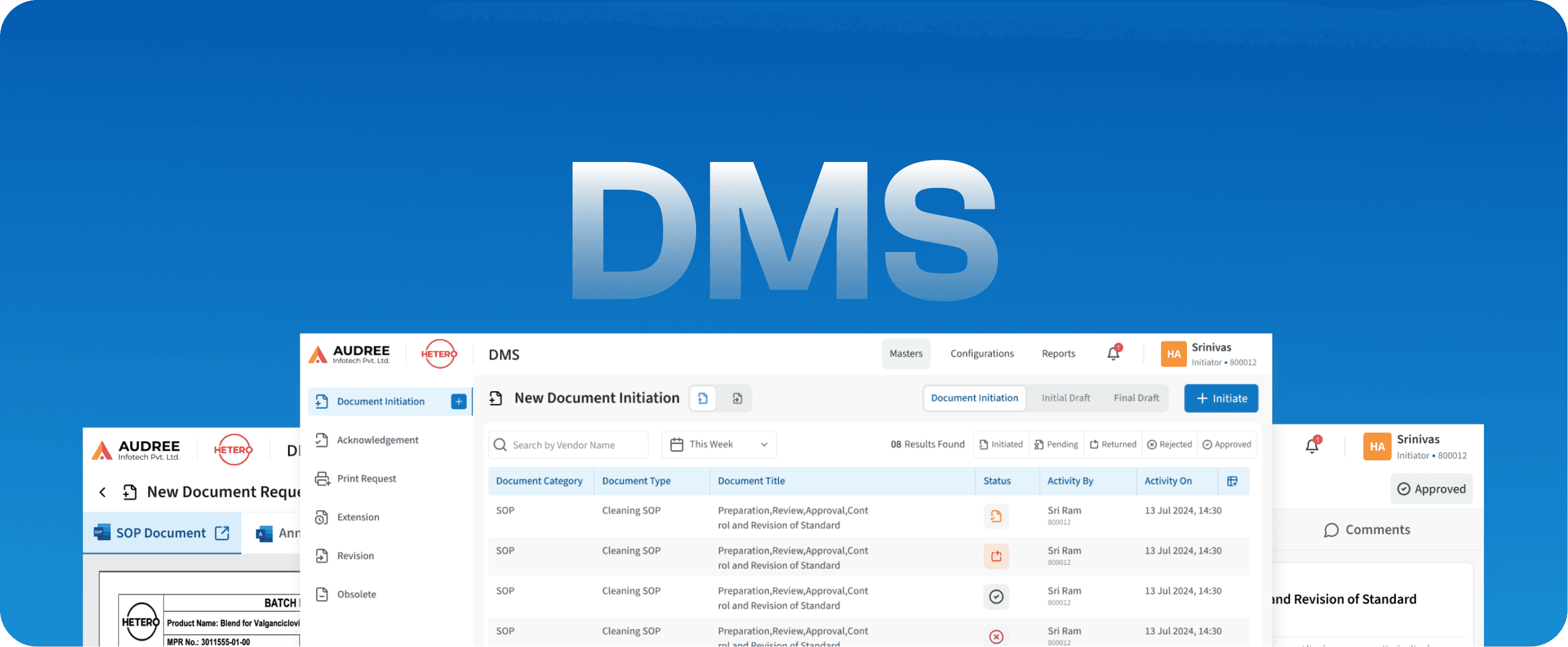
Document Management System
CAPA

Corrective And Preventive Actions
QAS

Quality Agreement System
Vendor Portal
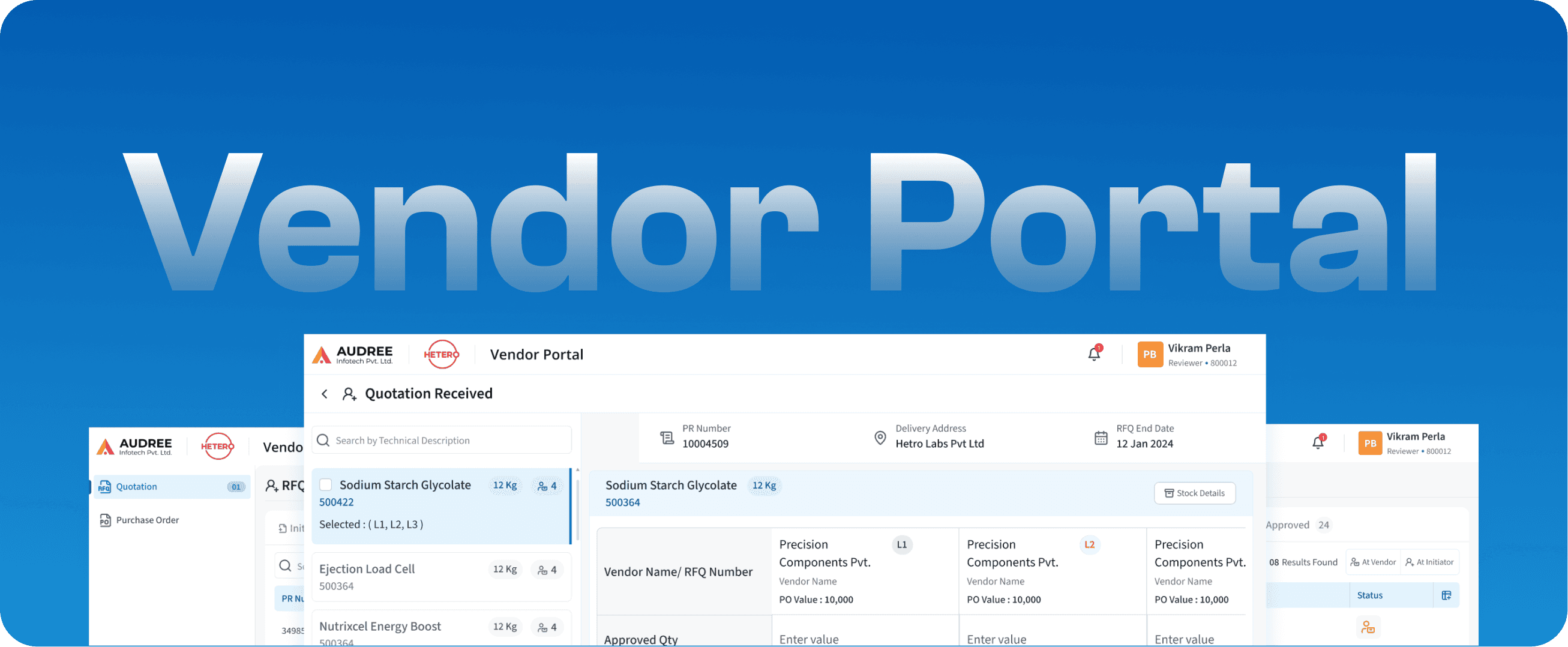
Vendor Management System
LIR-AER
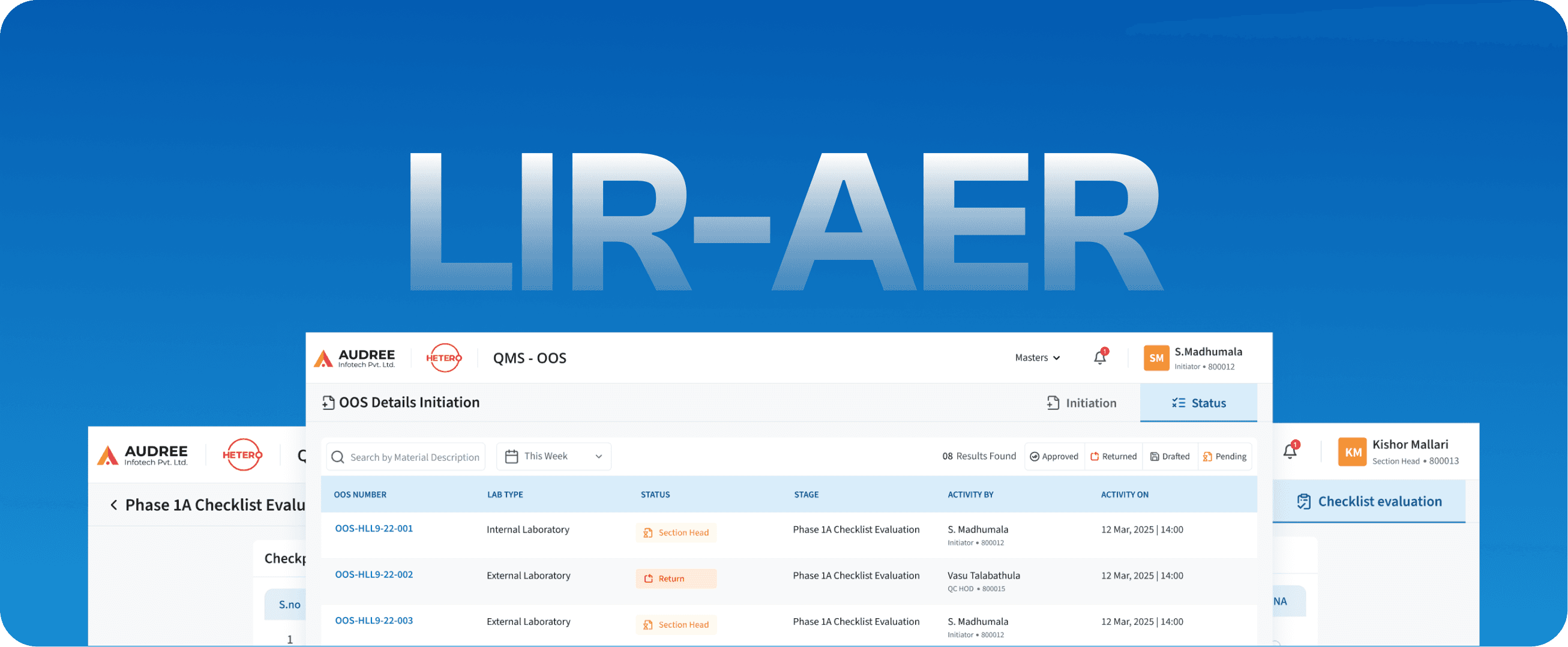
Laboratory Information Record
OOS
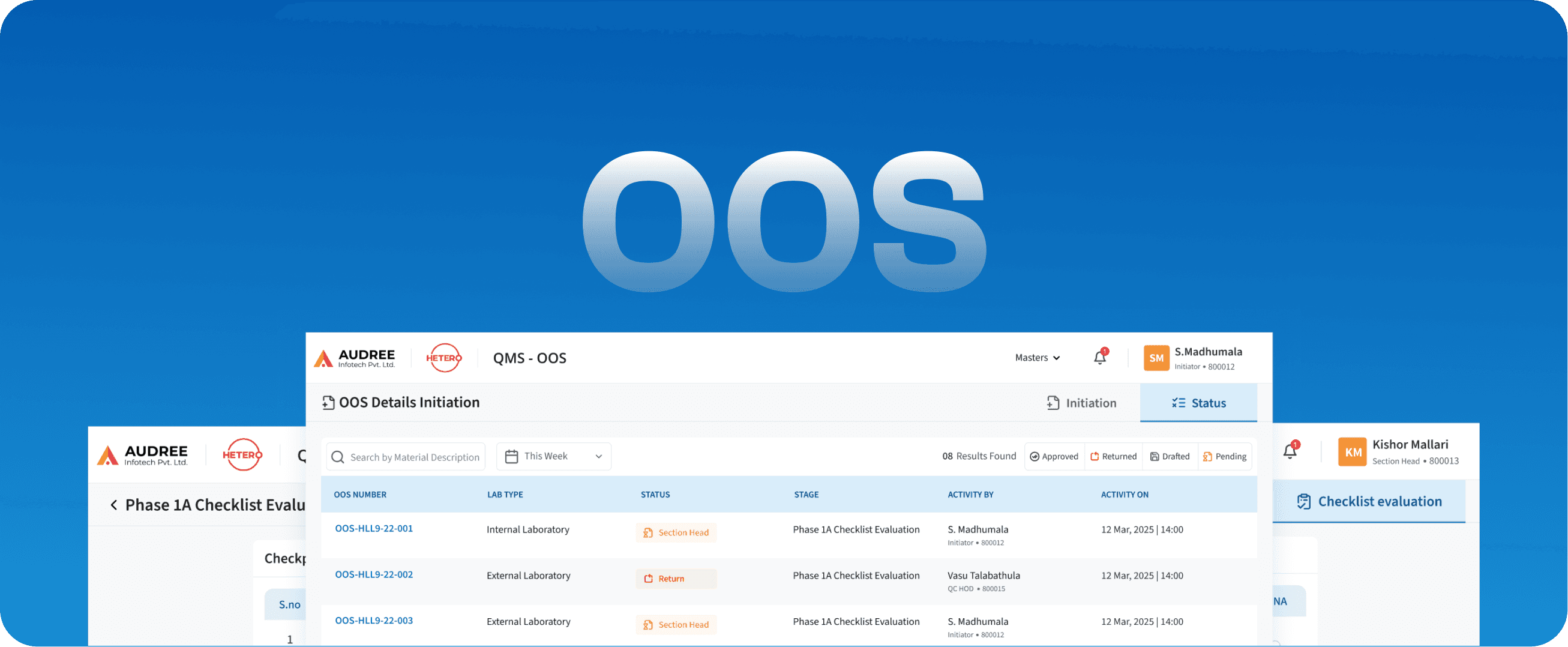
Out Of Specification
CMS
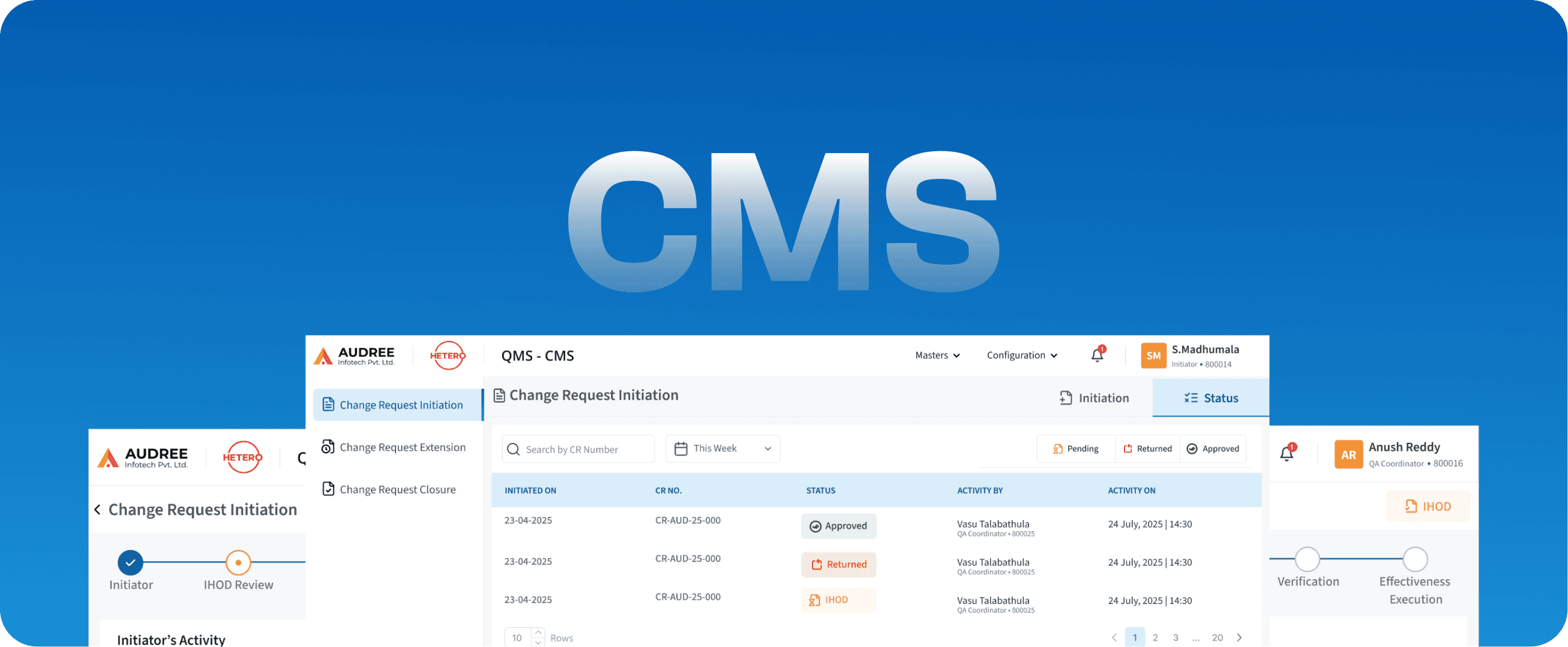
Change Management System
IMS
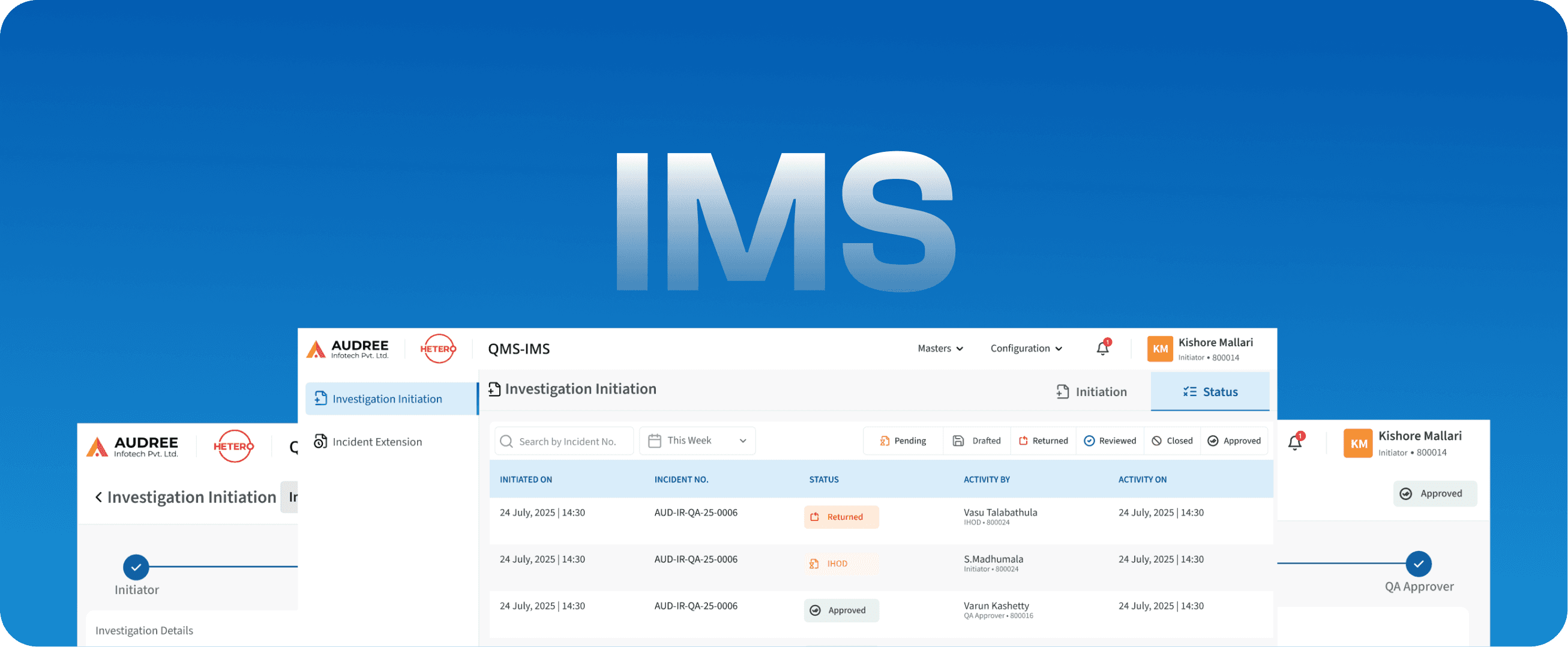
Incident Management System
BRMS-API
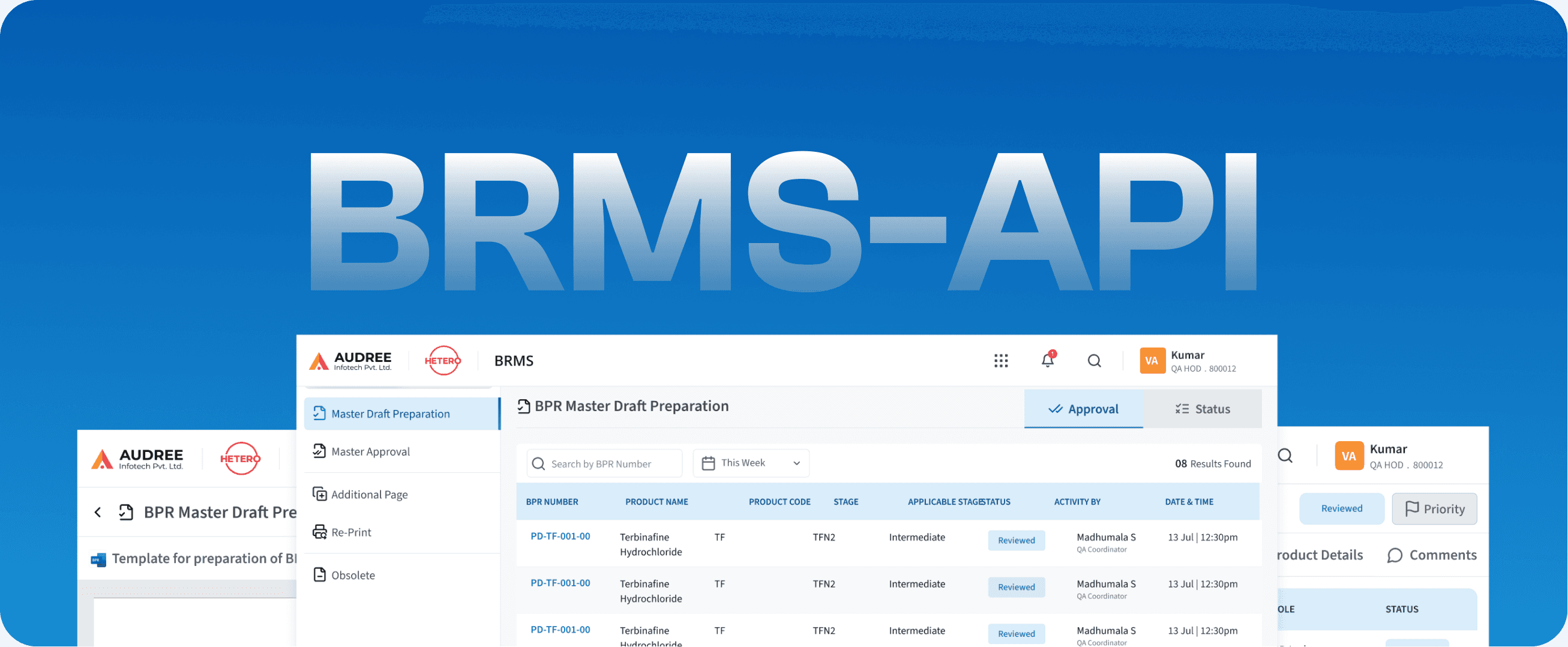
Batch Record- Active Pharmaceutical Ingredient
E-BRMS
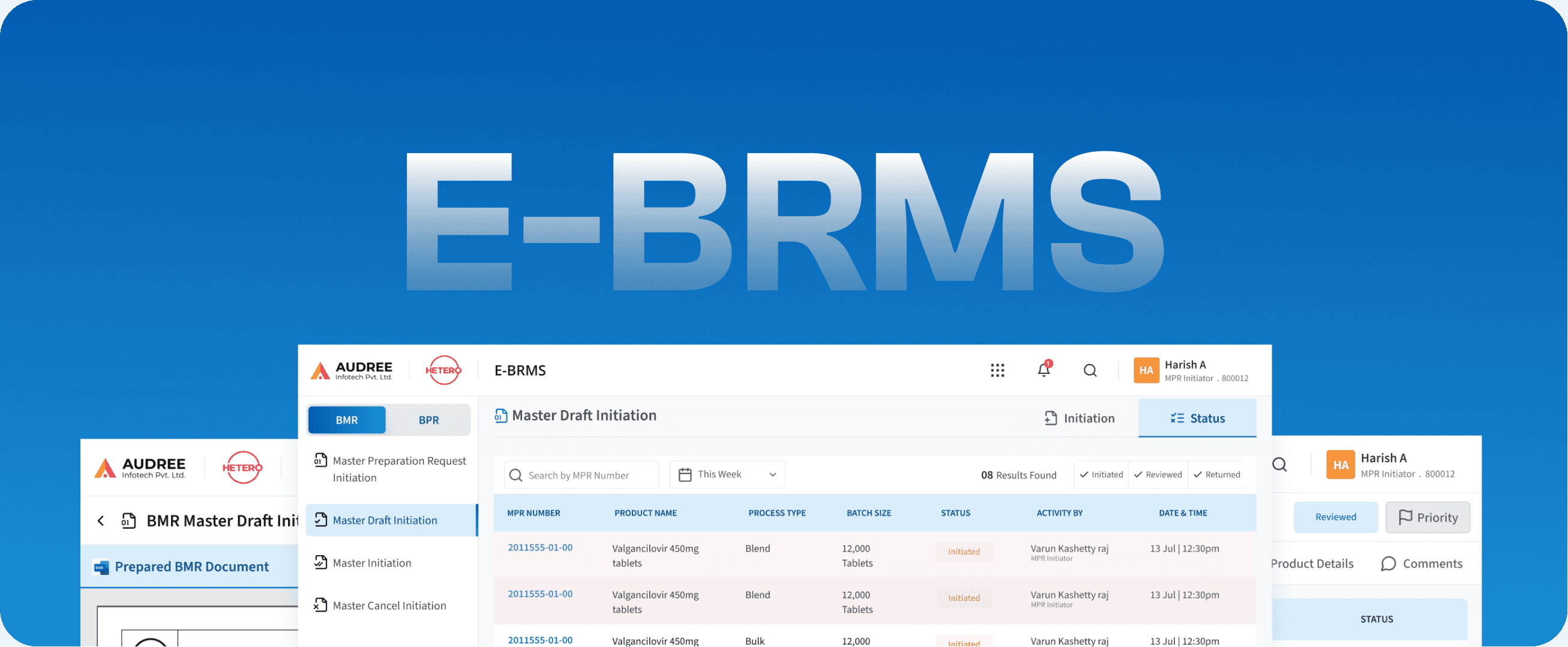
Batch Record Management System
RIMS
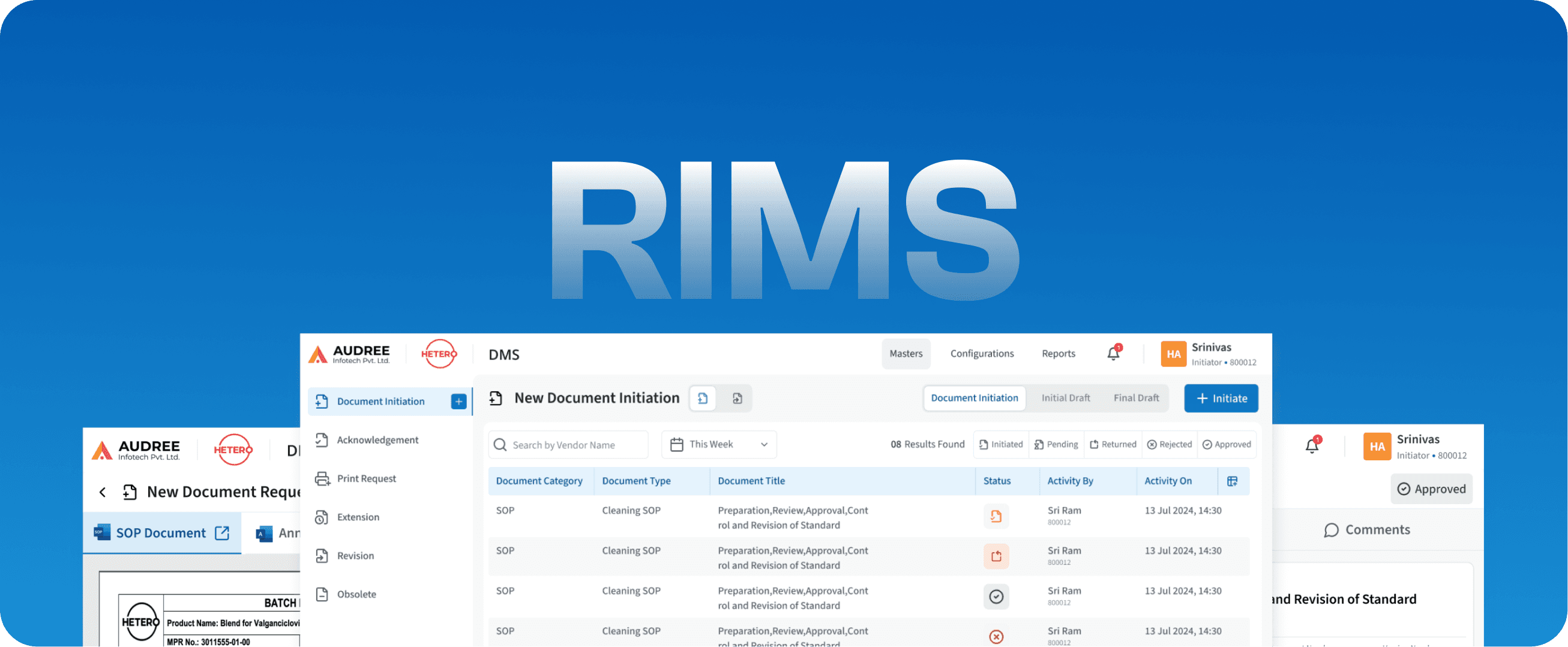
Regulatory Information Management System
APQR
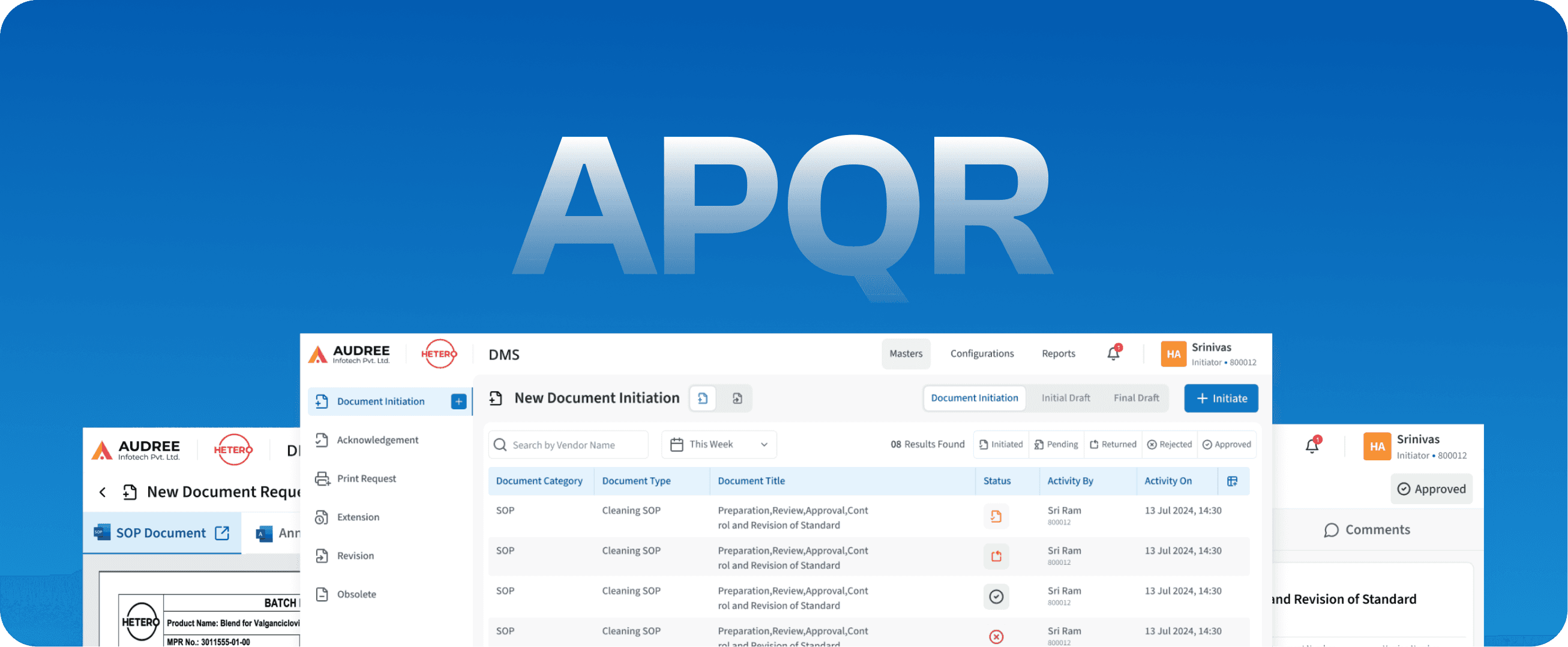
Annual Product Quality Review
RCAI

Root Cause Analysis with Intelligence
LMS

Learning Management System
LIMS

Laboratory Information Management System
S & OP

Sales & Operations Planning
E-BMR

Batch Manufacturing Recall
RIMS
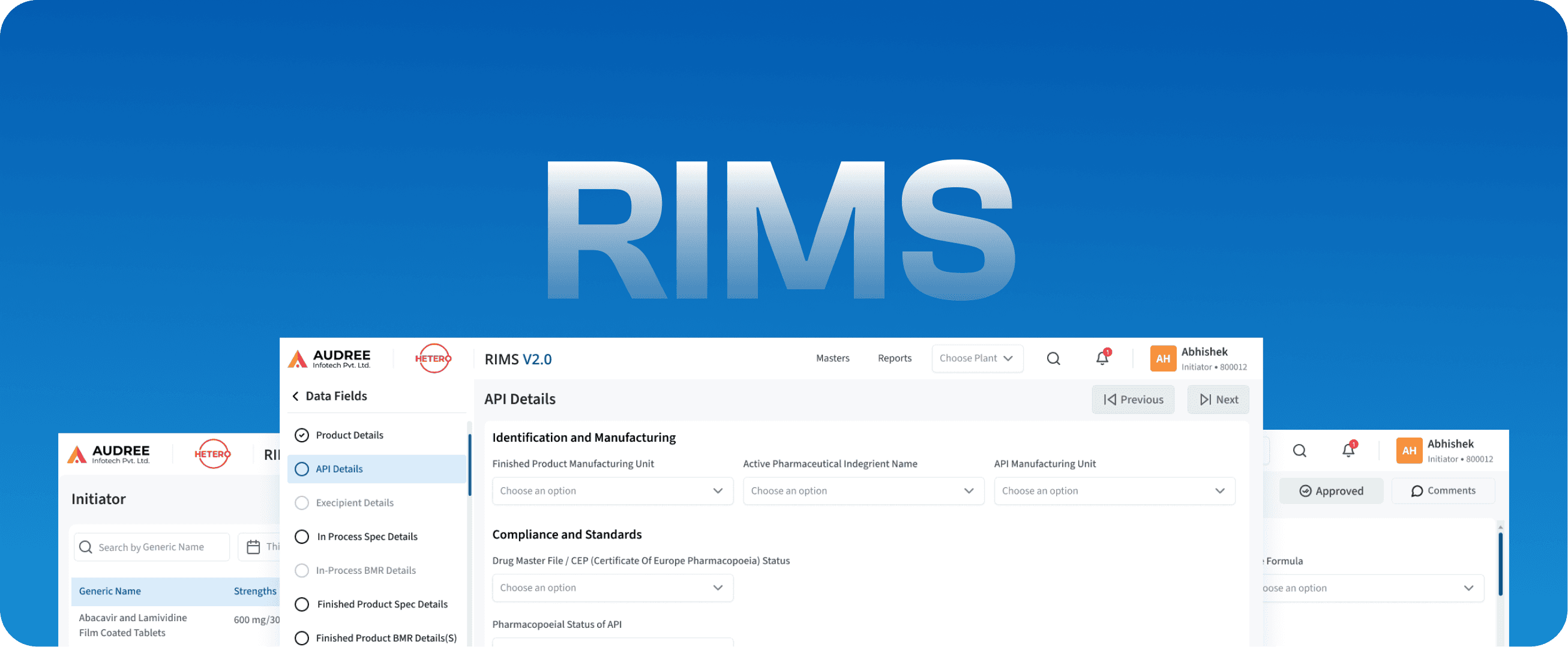
Regulatory Information Management Systems
IMS

Incident Management System
BRMS-API

Batch Record- Active Pharmaceutical Ingredient
E-BRMS

Batch Record Management System
APQR

Annual Product Quality Review
RIMS

Regulatory Information Management System
WMPS

Warehouse Management System
DMS

Document Management System
CMS

Change Management System
OOS

Out Of Specification
LIR-AER

Laboratory Information Record
Vendor Portal

Vendor Management System
QAS

Quality Agreement System
CAPA

Corrective And Preventive Actions

