Redesigning BMR Execution Into a Fully Digital, Plant-Ready Workflow
Redesigning BMR Execution Into a Fully Digital, Plant-Ready Workflow
Redesigning BMR Execution Into a Fully Digital, Plant-Ready Workflow
About Project
About Project
About Project
BMRs record every production step, ensuring traceability and compliance across the plant.
Initially, the digital BMR creation module was used by only 4 facilities in a single country.
After our redesign, its clarity, structure, and usability led to rapid adoption expanding to 36+ facilities across 4 countries worldwide. This success, Audree chose to collaborate with us again to digitise the execution phase, which was still dependent on paper logbooks, handwritten entries, and manual coordination.
The goal: a fully digital, end-to-end BMR lifecycle connecting creation → approval → execution → batch completion.
BMRs record every production step, ensuring traceability and compliance across the plant.
Initially, the digital BMR creation module was used by only 4 facilities in a single country.
After our redesign, its clarity, structure, and usability led to rapid adoption expanding to 36+ facilities across 4 countries worldwide. This success, Audree chose to collaborate with us again to digitise the execution phase, which was still dependent on paper logbooks, handwritten entries, and manual coordination.
The goal: a fully digital, end-to-end BMR lifecycle connecting creation → approval → execution → batch completion.
About Project
Pharmaceutical
Team
Anush Reddy, S.Madhumala
Subscription Category
Quick win
Project start Year
April 2025
Core Business Challenges
Core Business Challenges
Core Business Challenges
Poor User Adoption Due to Outdated Experience
Poor User Adoption Due to Outdated Experience
Poor User Adoption Due to Outdated Experience
Since execution remained fully paper-based, teams relied on logbooks and handwritten entries making the system hard to adopt and driving users to offline workarounds.
Since execution remained fully paper-based, teams relied on logbooks and handwritten entries making the system hard to adopt and driving users to offline workarounds.
Since execution remained fully paper-based, teams relied on logbooks and handwritten entries making the system hard to adopt and driving users to offline workarounds.
Stuck in a Loop, Slowed by Inefficiency
Stuck in a Loop, Slowed by Inefficiency
Stuck in a Loop, Slowed by Inefficiency
Paper-based execution, repeated entries, scattered logbooks, and recurring support tickets kept teams stuck in the same loops slowing batch completion.
Paper-based execution, repeated entries, scattered logbooks, and recurring support tickets kept teams stuck in the same loops slowing batch completion.
Paper-based execution, repeated entries, scattered logbooks, and recurring support tickets kept teams stuck in the same loops slowing batch completion.
Heavy Dependence on Manual Coordination
Heavy Dependence on Manual Coordination
Execution relied on calls, supervisor follow-ups, and manual verification, creating delays and increasing the risk of errors and inconsistencies.
Execution relied on calls, supervisor follow-ups, and manual verification, creating delays and increasing the risk of errors and inconsistencies.
Heavy Dependence on Manual Coordination
Execution relied on calls, supervisor follow-ups, and manual verification, creating delays and increasing the risk of errors and inconsistencies.
Our Approach
Our Approach
Our Approach
Mapping User Flows to Uncover Hidden Gaps
We mapped how BMRs actually move from drafting to on-floor execution, uncovering gaps in transitions, repeated steps, and manual coordination. This helped us design one continuous digital flow instead of disconnected stages.
Mapping User Flows to Uncover Hidden Gaps
We mapped how BMRs actually move from drafting to on-floor execution, uncovering gaps in transitions, repeated steps, and manual coordination. This helped us design one continuous digital flow instead of disconnected stages.
Mapping User Flows to Uncover Hidden Gaps
We mapped how BMRs actually move from drafting to on-floor execution, uncovering gaps in transitions, repeated steps, and manual coordination. This helped us design one continuous digital flow instead of disconnected stages.
Designing BMR Execution Into a Seamless Digital Workflow
Designing BMR Execution Into a Seamless Digital Workflow
Designing BMR Execution Into a Seamless Digital Workflow
We extended the BMR redesign from creation and approval to full on-floor execution, Execution was rebuilt into guided, stage-wise digital flows with clear instructions, validations, and role-based actions. With digital sign-offs and a unified Manufacturing–Packing view, teams no longer switch between PDFs or logbooks making execution faster, clearer, and fully traceable.
We extended the BMR redesign from creation and approval to full on-floor execution, Execution was rebuilt into guided, stage-wise digital flows with clear instructions, validations, and role-based actions. With digital sign-offs and a unified Manufacturing–Packing view, teams no longer switch between PDFs or logbooks making execution faster, clearer, and fully traceable.


Streamlined BMR Creation for Real Production Needs
Streamlined BMR Creation for Real Production Needs
Streamlined BMR Creation for Real Production Needs
We refined the BMR creation module by aligning it with the new execution workflow and using real production feedback. Stages were streamlined and statuses clarified, making the creation-to-issuance flow more structured and better connected.
We refined the BMR creation module by aligning it with the new execution workflow and using real production feedback. Stages were streamlined and statuses clarified, making the creation-to-issuance flow more structured and better connected.
We refined the BMR creation module by aligning it with the new execution workflow and using real production feedback. Stages were streamlined and statuses clarified, making the creation-to-issuance flow more structured and better connected.




Turning Paper-Heavy Execution Into a Real-Time Digital Workflow
Turning Paper-Heavy Execution Into a Real-Time Digital Workflow
Turning Paper-Heavy Execution Into a Real-Time Digital Workflow
We digitised the entire shop-floor execution flow with guided steps, digital checklists, and real-time prompts that match actual manufacturing sequences. Paper logbooks and offline verification were replaced with actionable tasks, built-in validations, equipment mapping, and digital sign-offs all reducing errors and speeding up execution. role-based views, and instant QA visibility ensure smoother operations and faster batch closure.
We digitised the entire shop-floor execution flow with guided steps, digital checklists, and real-time prompts that match actual manufacturing sequences. Paper logbooks and offline verification were replaced with actionable tasks, built-in validations, equipment mapping, and digital sign-offs all reducing errors and speeding up execution. role-based views, and instant QA visibility ensure smoother operations and faster batch closure.
We digitised the entire shop-floor execution flow with guided steps, digital checklists, and real-time prompts that match actual manufacturing sequences. Paper logbooks and offline verification were replaced with actionable tasks, built-in validations, equipment mapping, and digital sign-offs all reducing errors and speeding up execution. role-based views, and instant QA visibility ensure smoother operations and faster batch closure.



Production Steps Made Instantly Understandable
Production Steps Made Instantly Understandable
Production Steps Made Instantly Understandable
Progress indicators make each stage of execution easy to follow. Operators instantly know what’s completed, what’s in progress, and what’s next reducing confusion and ensuring a clarity.
Progress indicators make each stage of execution easy to follow. Operators instantly know what’s completed, what’s in progress, and what’s next reducing confusion and ensuring a clarity.






A Modern UI for Faster, Cleaner Batch Records
A Modern UI for Faster, Cleaner Batch Records
A Modern UI for Faster, Cleaner Batch Records
Every screen is built for clarity clean tables, guided actions, and visual indicators help users move faster, reduce errors, and stay aligned across production and QA.
Every screen is built for clarity clean tables, guided actions, and visual indicators help users move faster, reduce errors, and stay aligned across production and QA.



Results That Transformed BMR Lifecycle
Results That Transformed BMR Lifecycle
Results That Transformed BMR Lifecycle
Clearer workflows, connected stages, and a fully digital execution process accelerated manufacturing, reduced errors, and strengthened traceability across every plant.
Clearer workflows, connected stages, and a fully digital execution process accelerated manufacturing, reduced errors, and strengthened traceability across every plant.

Stronger Compliance & Traceability
Stronger Compliance & Traceability
Stronger Compliance & Traceability
Real-time QA visibility, instant e-signatures, automated logs, and structured checklists reduced errors and rework. Each action is now fully traceable, improving audit readiness and reducing compliance gaps.
Real-time QA visibility, instant e-signatures, automated logs, and structured checklists reduced errors and rework. Each action is now fully traceable, improving audit readiness and reducing compliance gaps.
Real-time QA visibility, instant e-signatures, automated logs, and structured checklists reduced errors and rework. Each action is now fully traceable, improving audit readiness and reducing compliance gaps.

Faster API Batch Execution
Streamlined digital flows replaced paper logbooks and manual entries, enabling operators to move through each stage quickly with guided steps and automated validations. This drastically shortened execution time across manufacturing lines.

Reduced Follow-ups & Support Dependency
Unified records, clearer stage visibility, and predictable version-controlled updates reduced user confusion and dependency on support teams. Teams no longer chase updates every detail is visible in the flow itself.

Faster API Batch Execution
Faster API Batch Execution
Streamlined digital flows replaced paper logbooks and manual entries, enabling operators to move through each stage quickly with guided steps and automated validations. This drastically shortened execution time across manufacturing lines.
Streamlined digital flows replaced paper logbooks and manual entries, enabling operators to move through each stage quickly with guided steps and automated validations. This drastically shortened execution time across manufacturing lines.

Reduced Follow-ups & Support Dependency
Reduce Follow-ups & Support Dependency
Unified records, clearer stage visibility, and predictable version-controlled updates reduced user confusion and dependency on support teams. Teams no longer chase updates every detail is visible in the flow itself.
Unified records, clearer stage visibility, and predictable version-controlled updates reduced user confusion and dependency on support teams. Teams no longer chase updates every detail is visible in the flow itself.
Deep-Dive Into More System
Deep-Dive Into More System
Deep-Dive Into More System
Browse every optimised Software and explore how legacy systems became intuitive.
Browse every optimised Software and explore how legacy systems became intuitive.
Browse every optimised Software and explore how legacy systems became intuitive.
RCAI

Root Cause Analysis with Intelligence
LMS
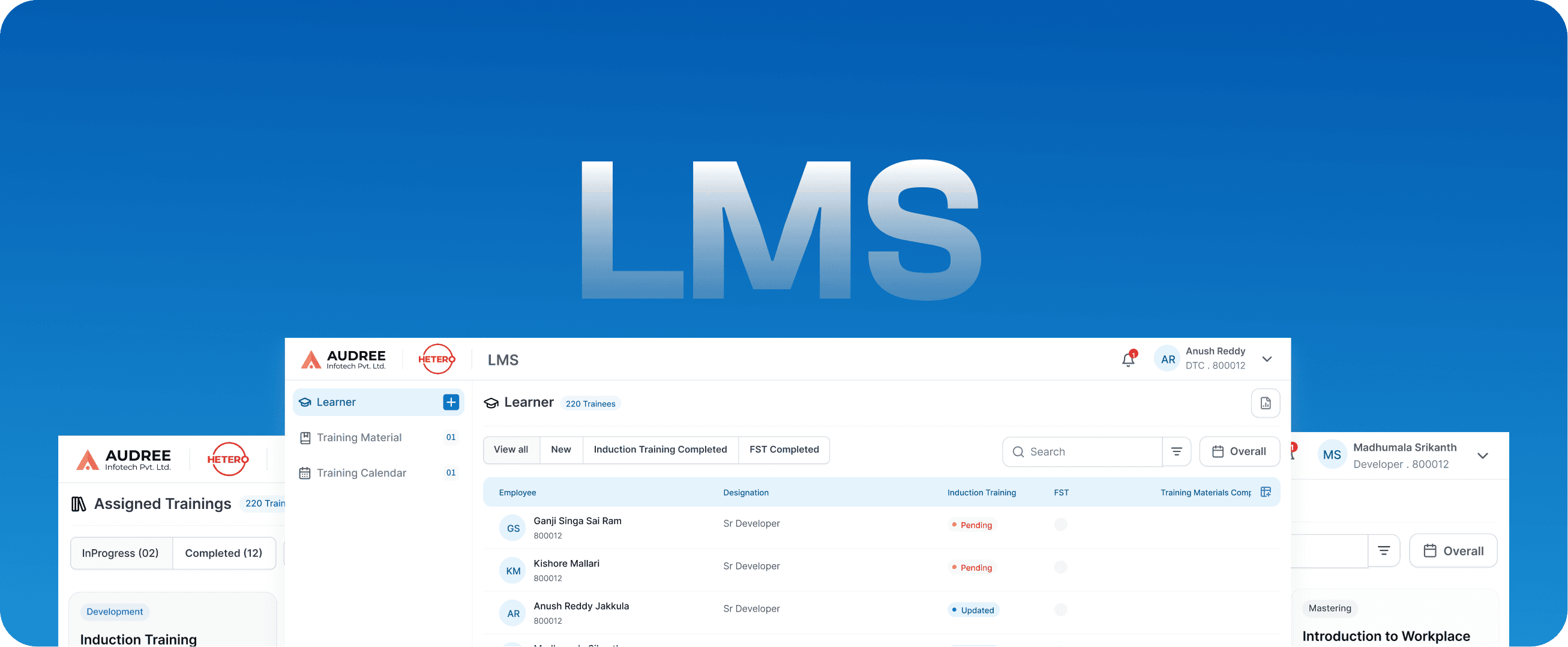
Learning Management System
LIMS
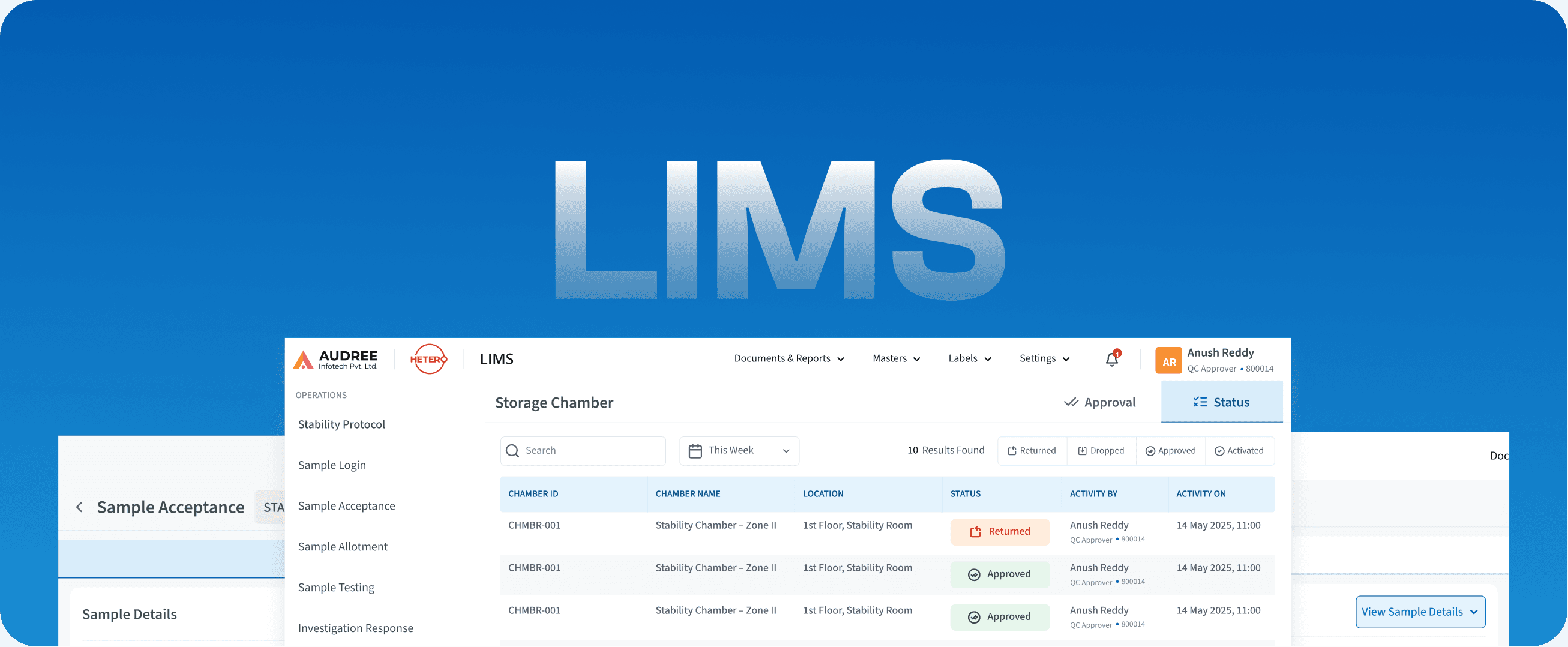
Laboratory Information Management System
S & OP
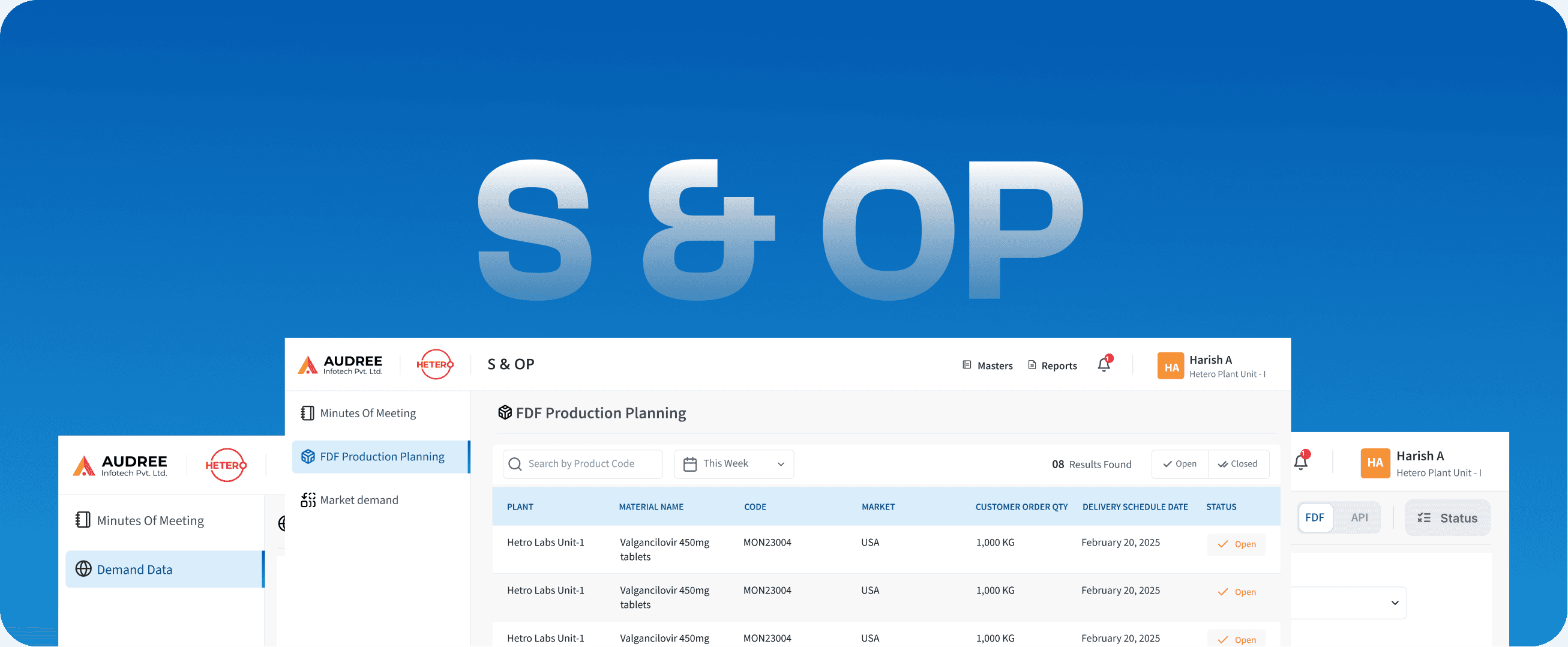
Sales & Operations Planning
E-BMR
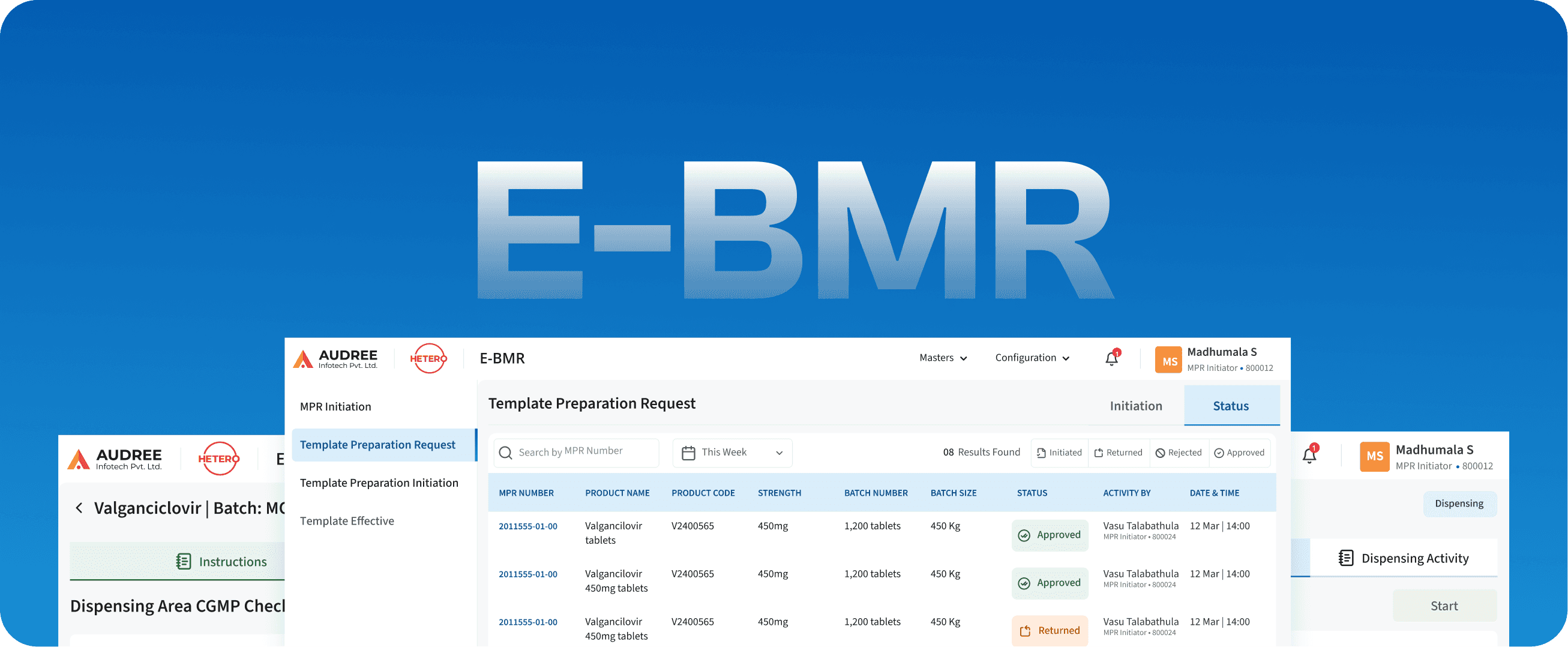
Batch Manufacturing Recall
WMPS

Warehouse Management System
DMS
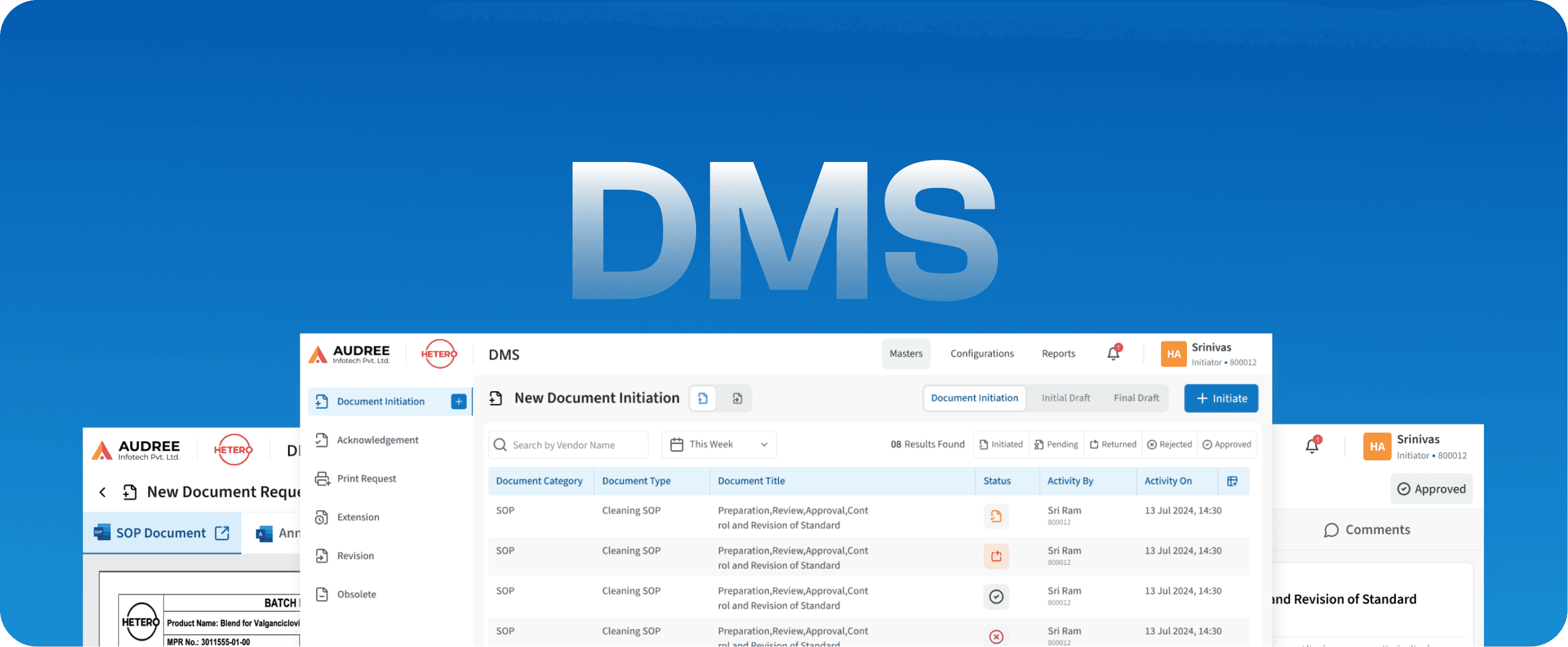
Document Management System
CAPA

Corrective And Preventive Actions
QAS

Quality Agreement System
Vendor Portal
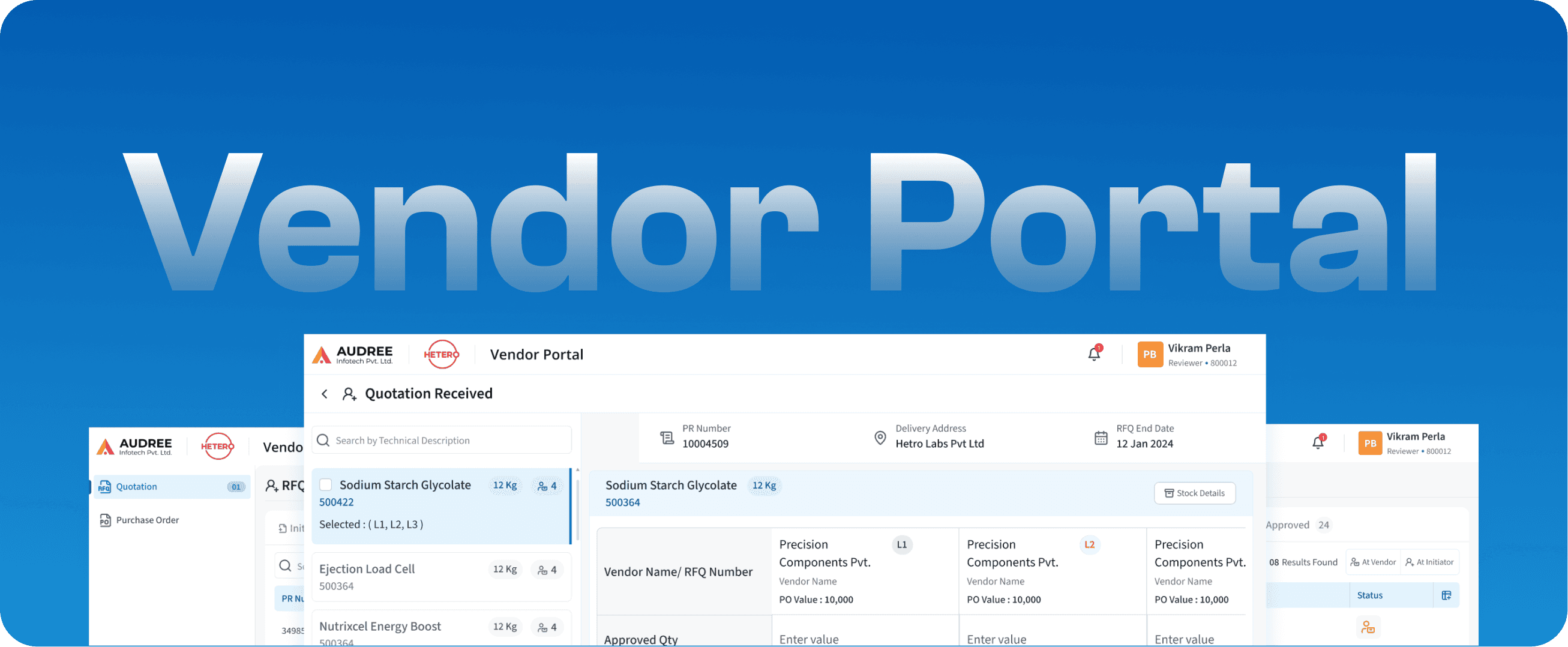
Vendor Management System
LIR-AER
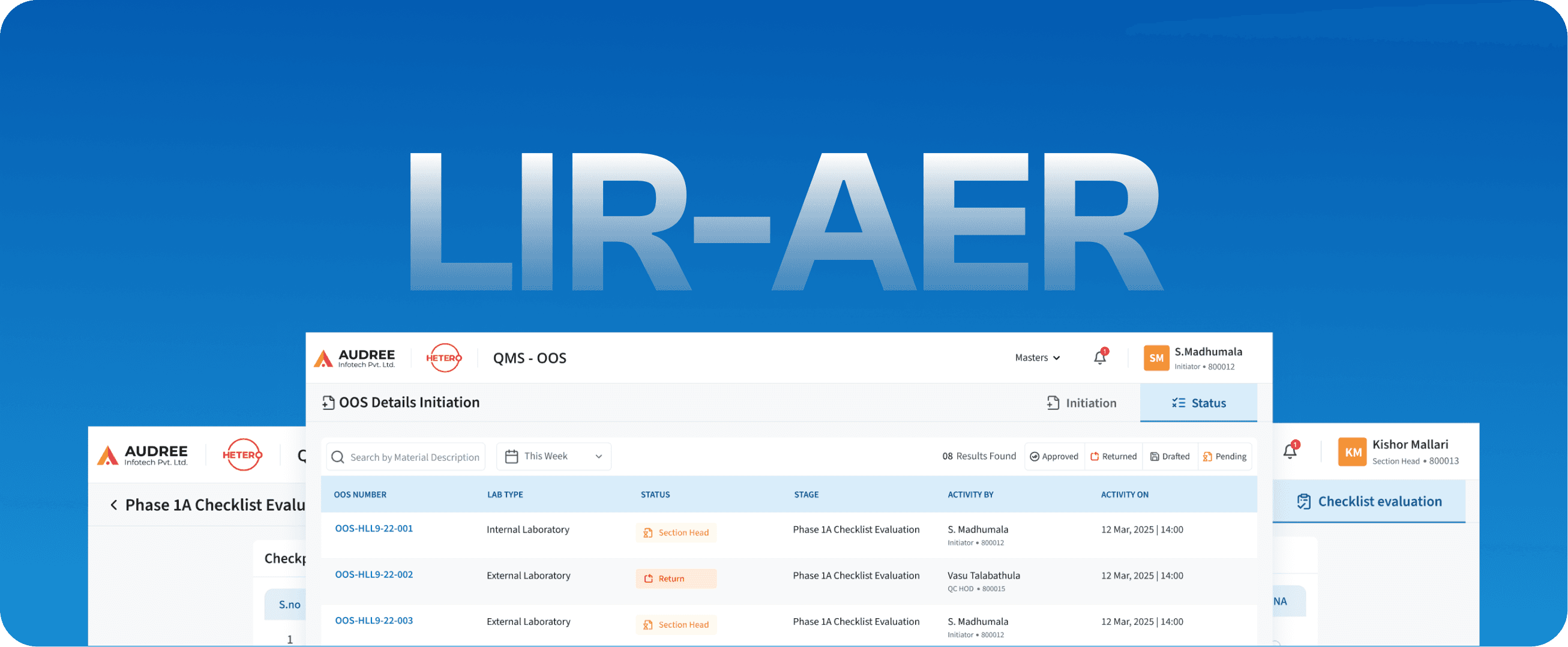
Laboratory Information Record
OOS
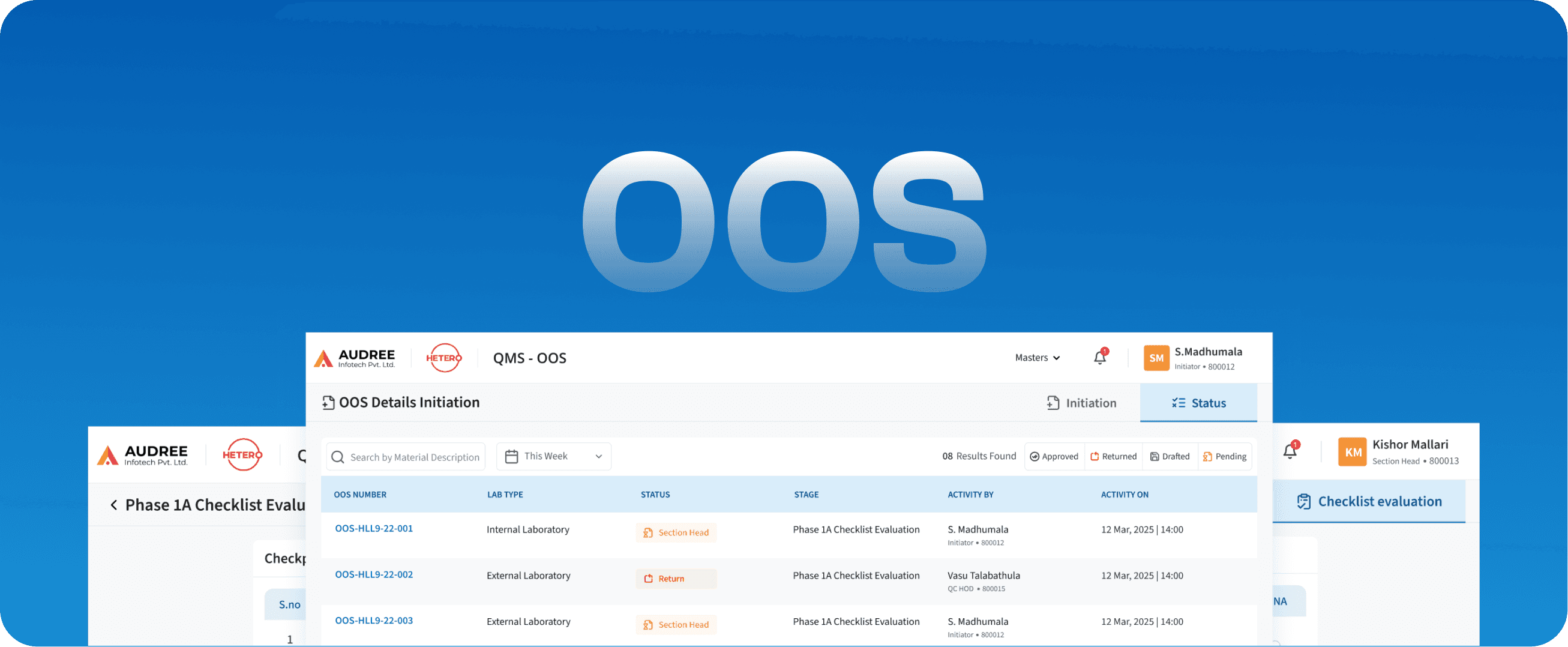
Out Of Specification
CMS
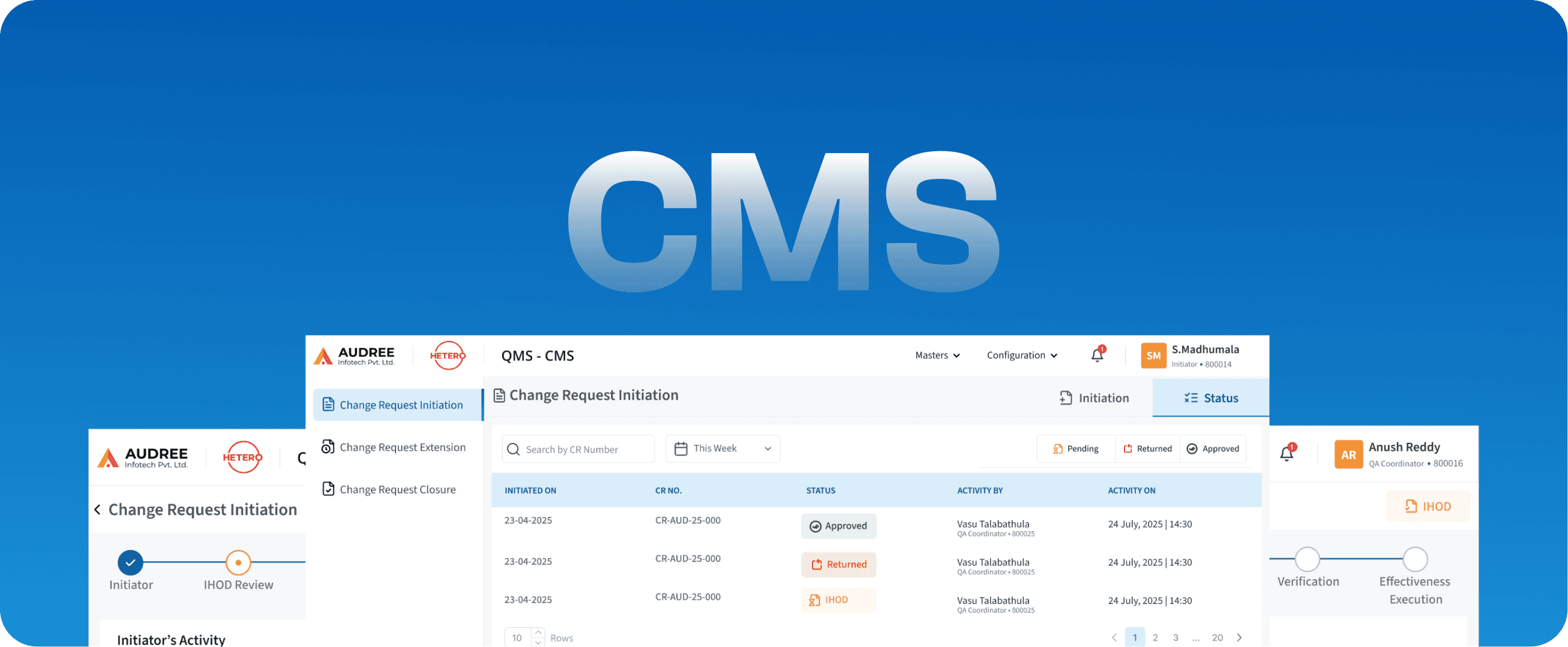
Change Management System
IMS
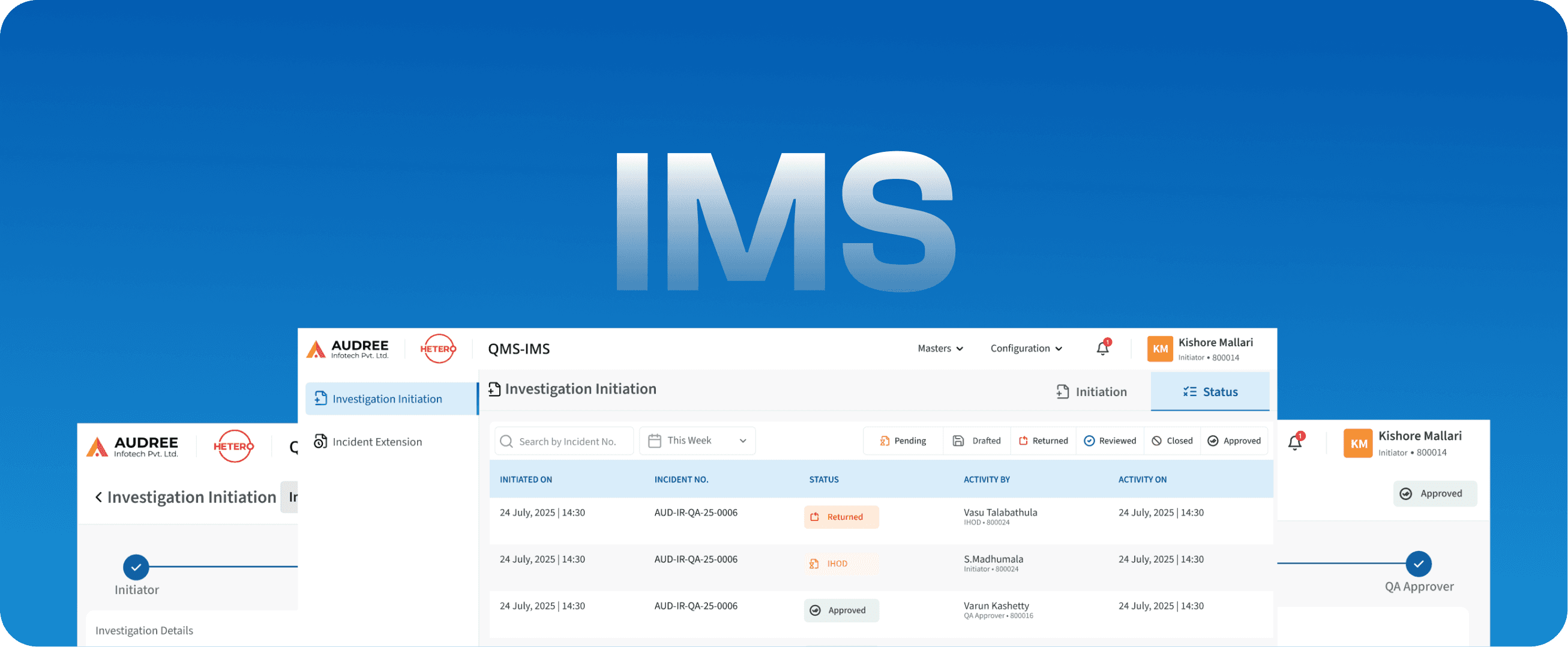
Incident Management System
BRMS-API
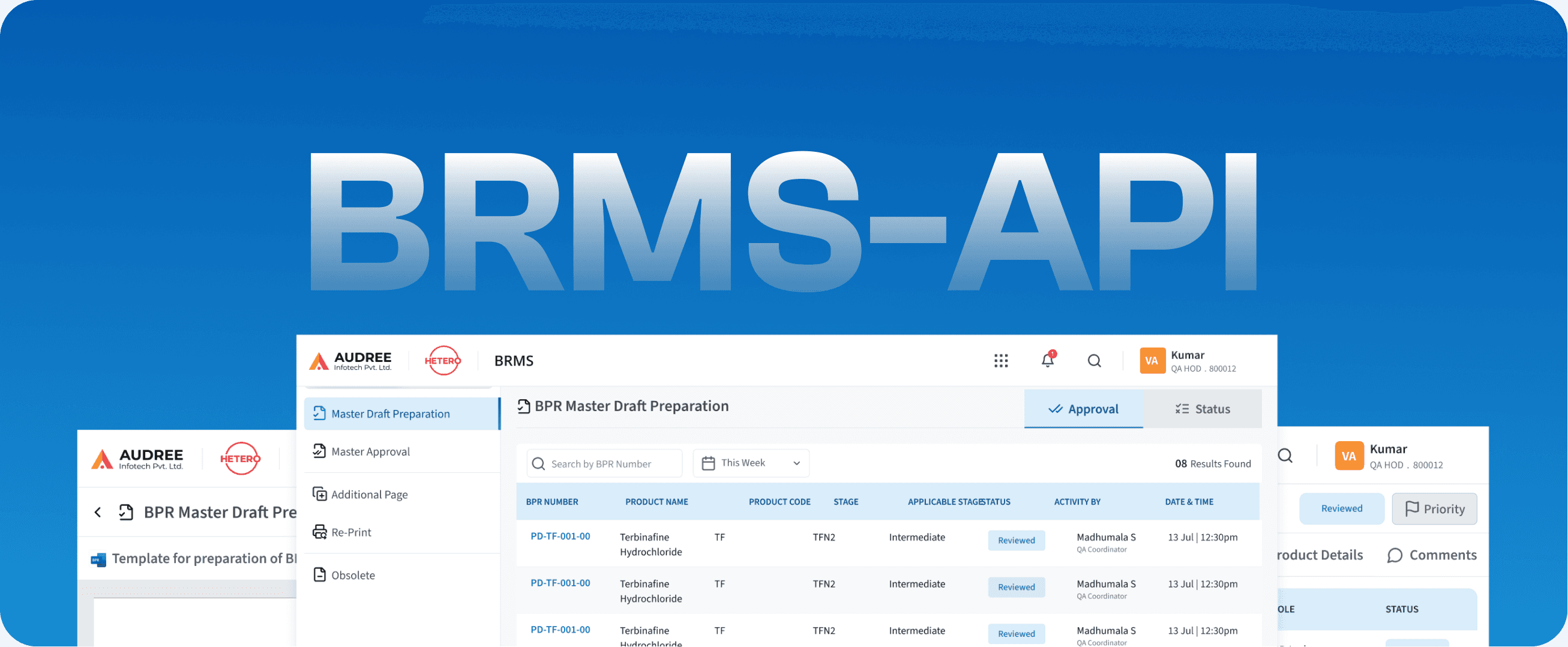
Batch Record- Active Pharmaceutical Ingredient
E-BRMS
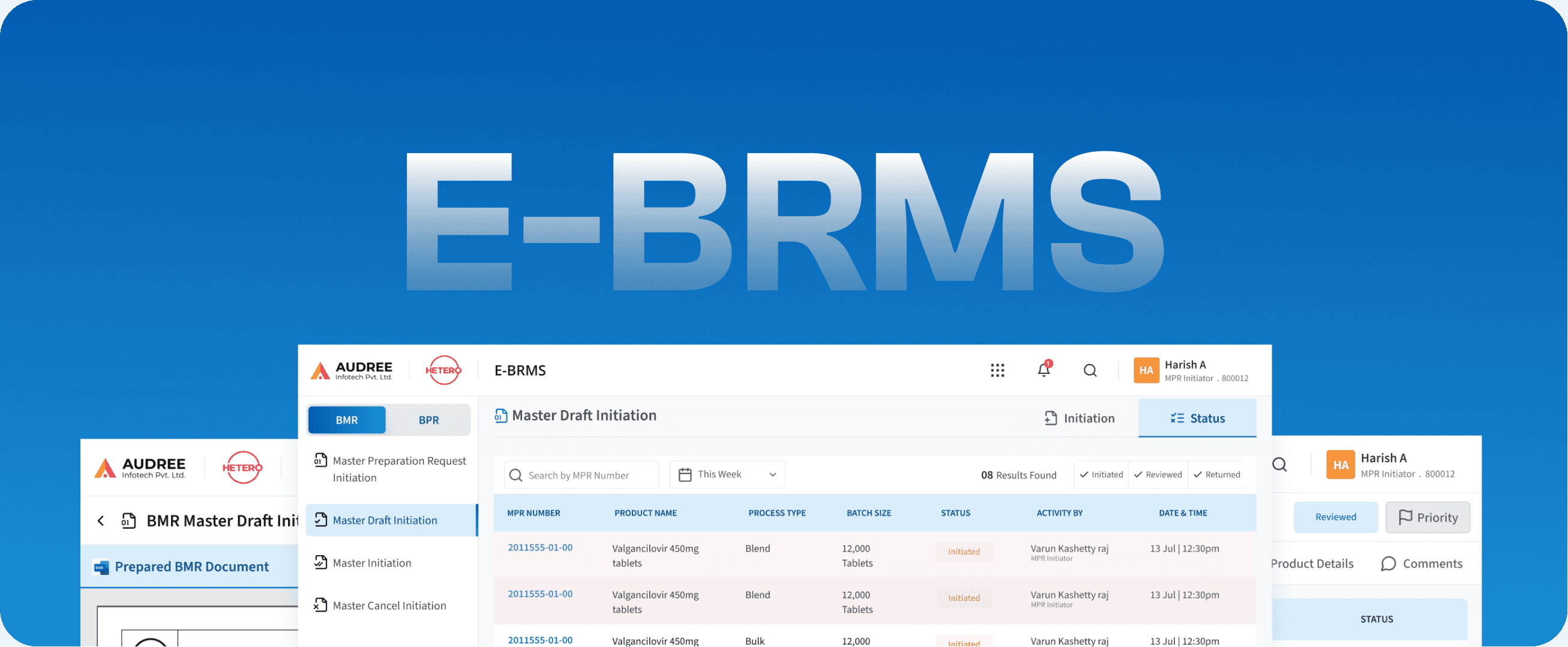
Batch Record Management System
RIMS
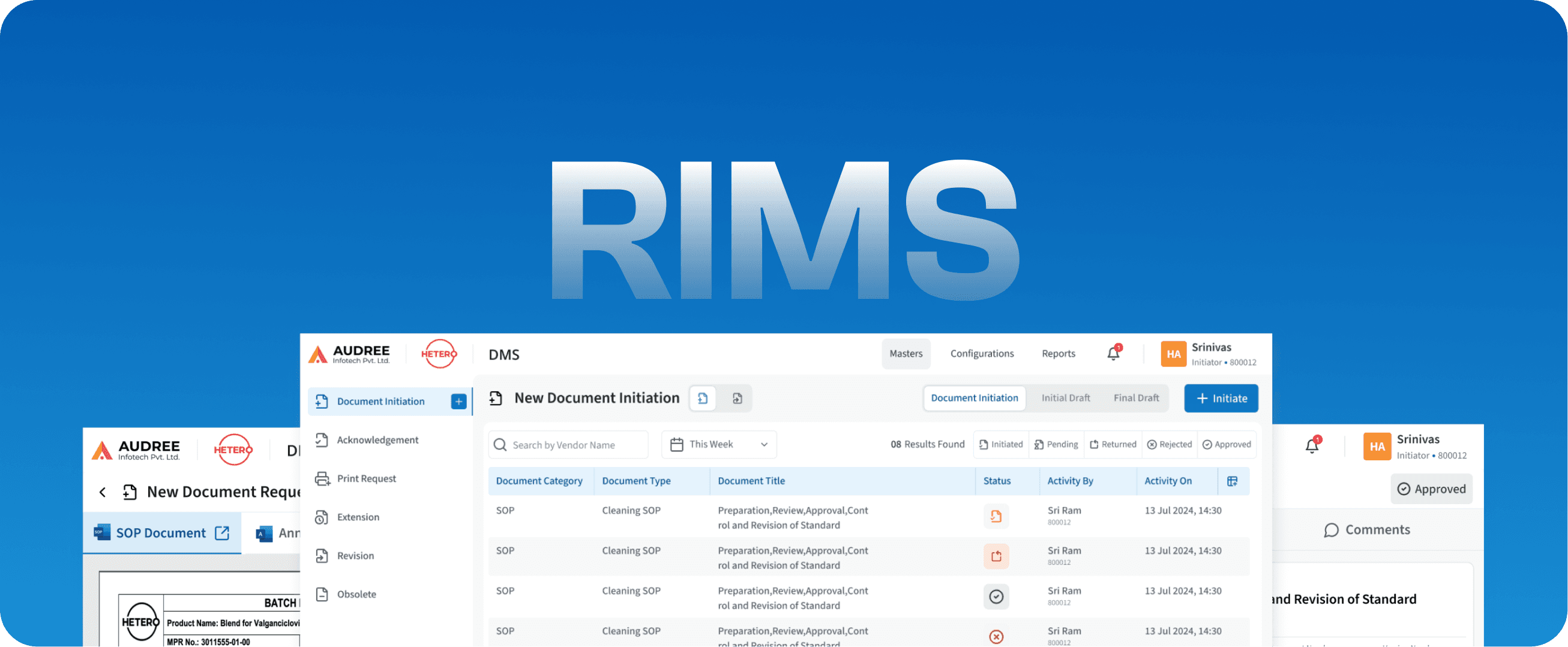
Regulatory Information Management System
APQR
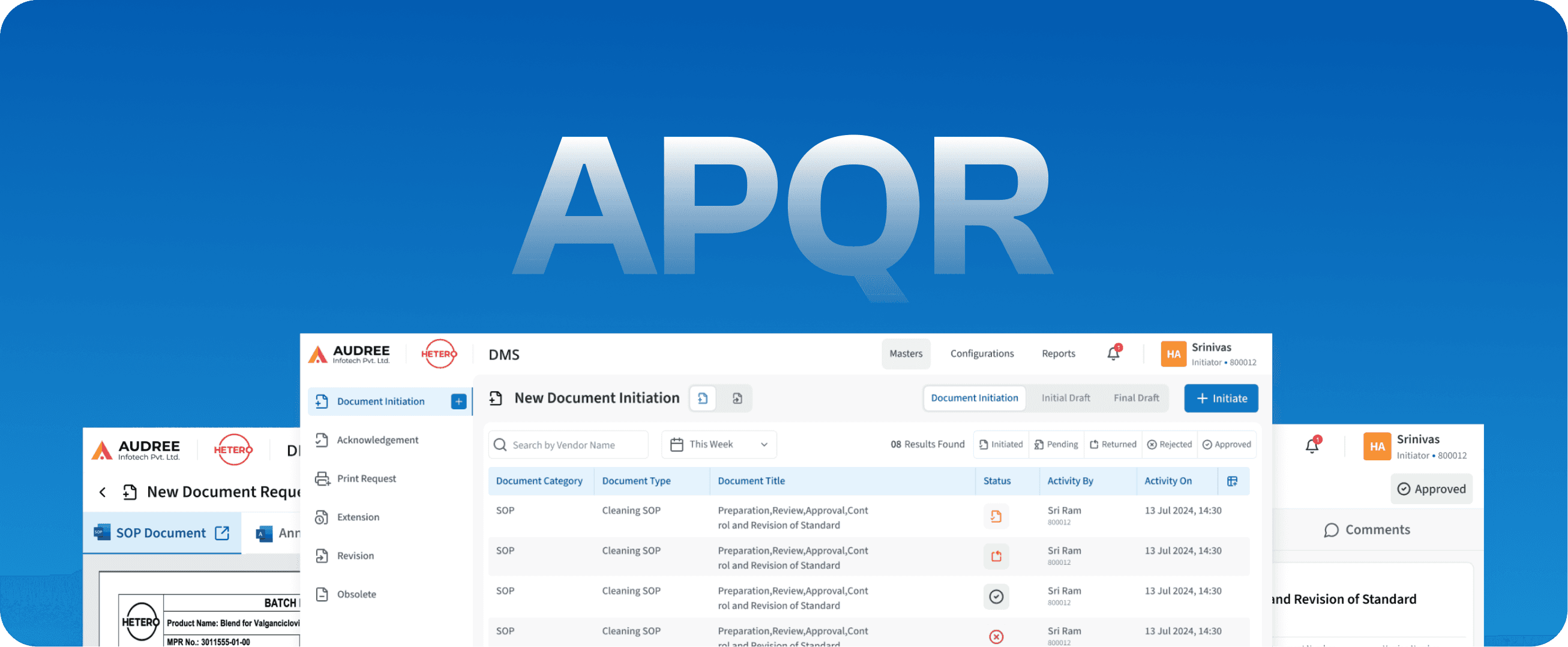
Annual Product Quality Review
RCAI

Root Cause Analysis with Intelligence
LMS

Learning Management System
LIMS

Laboratory Information Management System
S & OP

Sales & Operations Planning
E-BMR

Batch Manufacturing Recall
RIMS
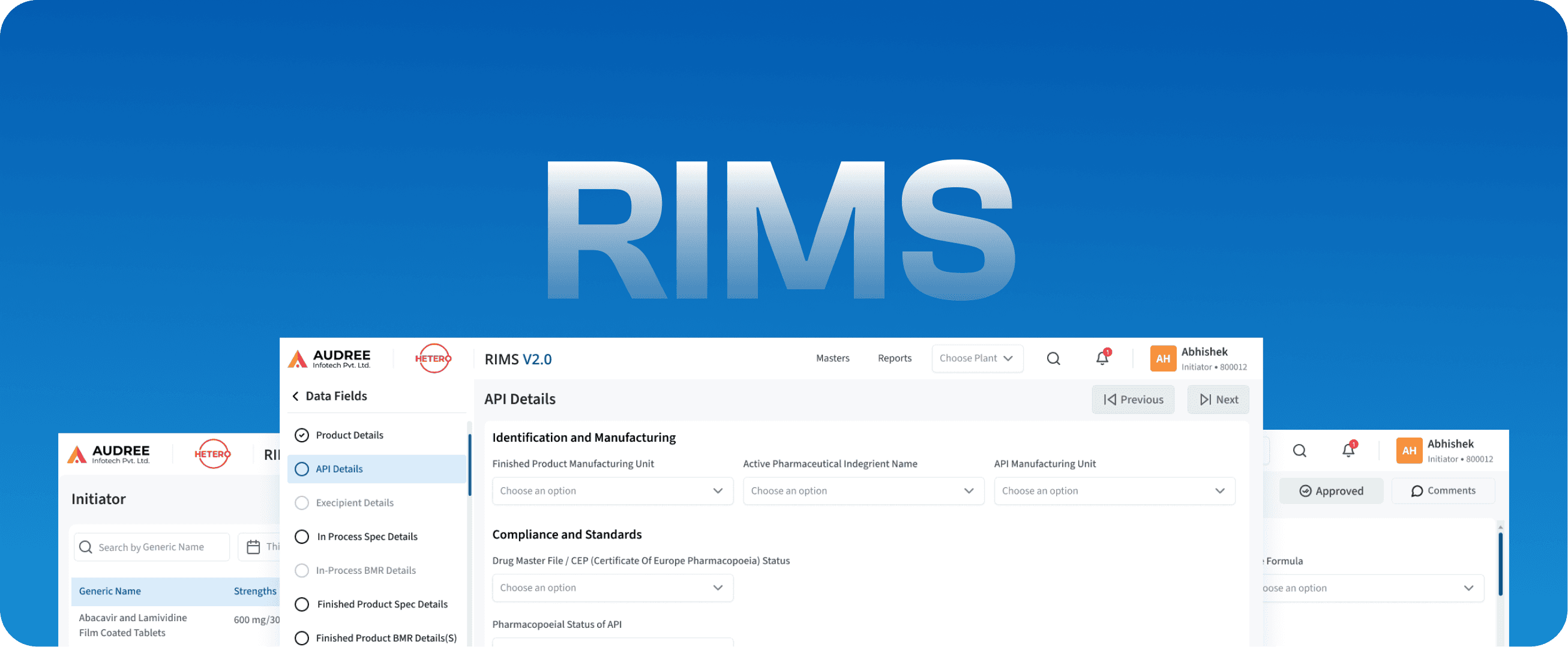
Regulatory Information Management Systems
IMS

Incident Management System
BRMS-API

Batch Record- Active Pharmaceutical Ingredient
E-BRMS

Batch Record Management System
APQR

Annual Product Quality Review
RIMS

Regulatory Information Management System
WMPS

Warehouse Management System
DMS

Document Management System
CMS

Change Management System
OOS

Out Of Specification
LIR-AER

Laboratory Information Record
Vendor Portal

Vendor Management System
QAS

Quality Agreement System
CAPA

Corrective And Preventive Actions

